Search Thermo Fisher Scientific
Invitrogen
O-GlcNAc Monoclonal Antibody (RL2), PE-Cyanine7, eBioscience™
This Antibody was verified by Cell treatment to ensure that the antibody binds to the antigen stated.
FIGURE: 1 / 3
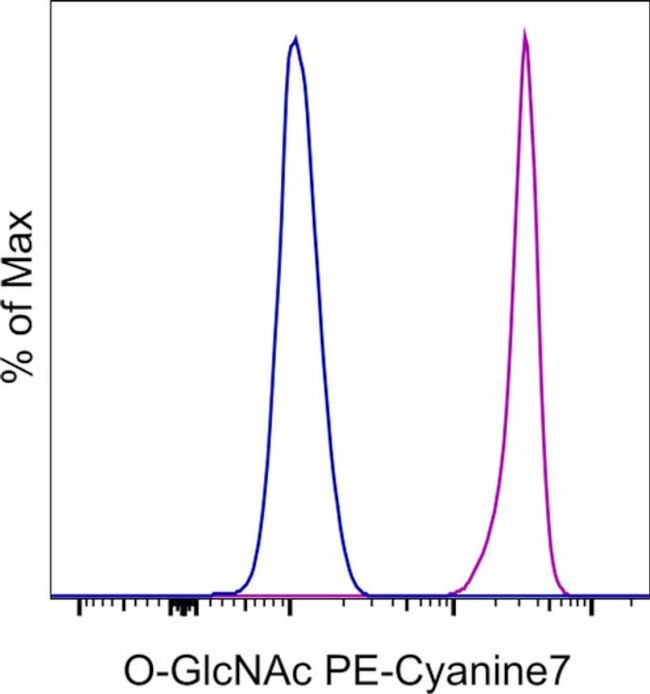
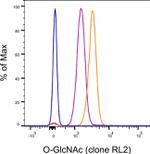
O-GlcNAc Antibody (25-9793-42) in Flow

Product Details
25-9793-42
Species Reactivity
Host/Isotype
Recommended Isotype Control
Class
Type
Clone
Immunogen
Conjugate
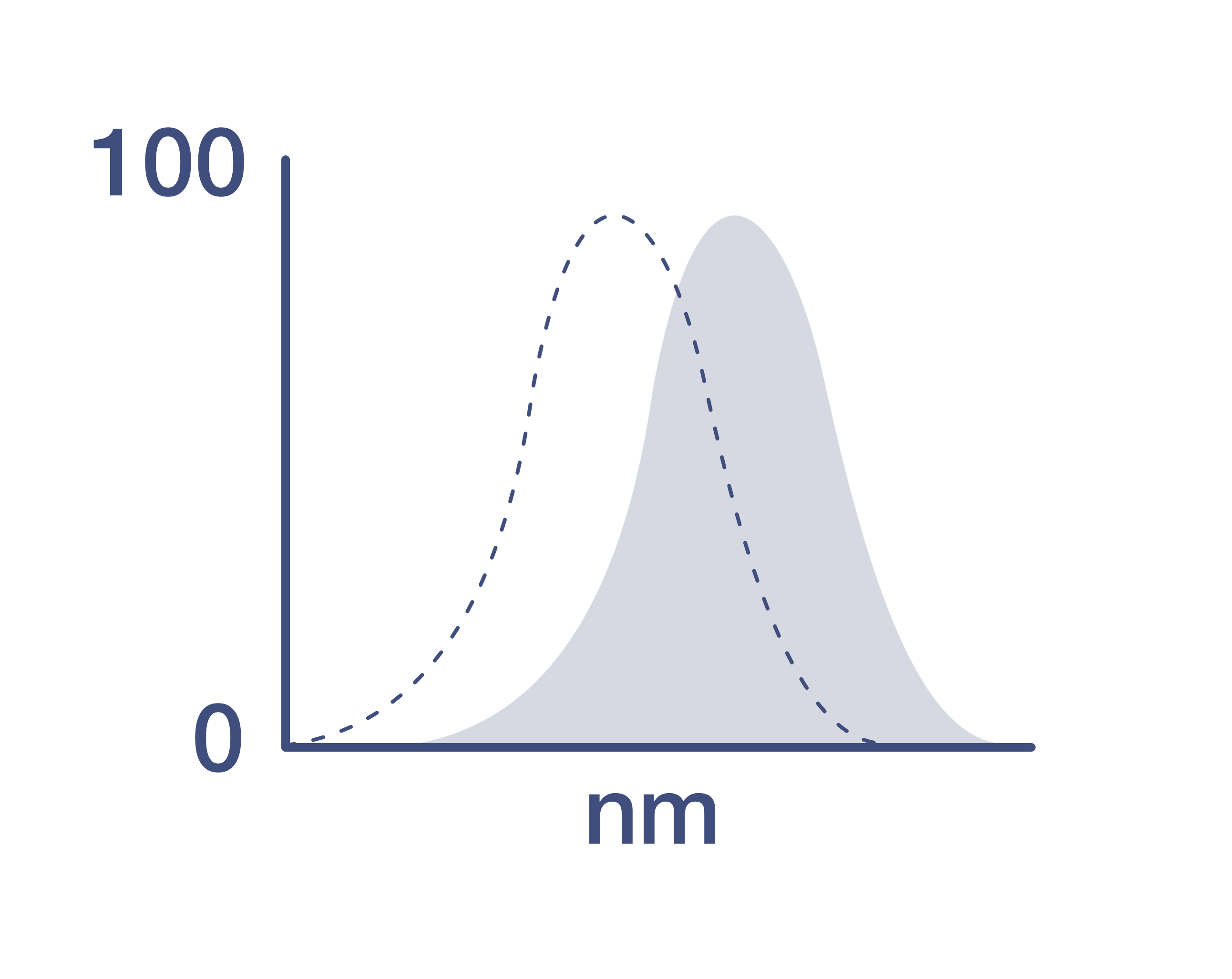
Excitation/Emission Max
Form
Concentration
Purification
Storage buffer
Contains
Storage conditions
Shipping conditions
RRID
Product Specific Information
Description: This RL2 monoclonal antibody recognizes proteins with O-N-acetylglucosamine (O-GlcNAc) glycosylation. It was originally developed by immunizing mice with the rat liver nuclear envelopes containing nuclear pore complexes. The RL2 clone has been successfully used in Western blot, immunofluorescence, immunoprecipitation, and flow cytometry in a wide variety of mammalian cells.
Applications Reported: This RL2 antibody has been reported for use in flow cytometric analysis.
Applications Tested: This RL2 antibody has been pre-diluted and tested by flow cytometric analysis of normal human peripheral blood cells using the Foxp3/Transcription Factor Staining Buffer Set (Product # 00-5523) and protocol. Please refer to Best Protocols: Protocol B: One step protocol for (nuclear) intracellular proteins located under the Resources Tab online. This may be used at 5 µL (0.25 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test.
Light sensitivity: This tandem dye is sensitive to photo-induced oxidation. Please protect this vial and stained samples from light.
Fixation: Samples can be stored in IC Fixation Buffer (Product # 00-8222) (100 µL of cell sample + 100 µL of IC Fixation Buffer) or 1-step Fix/Lyse Solution (Product # 00-5333) for up to 3 days in the dark at 4°C with minimal impact on brightness and FRET efficiency/compensation. Some generalizations regarding fluorophore performance after fixation can be made, but clone specific performance should be determined empirically.
Excitation: 488-561 nm; Emission: 775 nm; Laser: Blue Laser, Green Laser, Yellow-Green Laser
Target Information
O-linked N-acetylglucosamine (O-GlcNAc) is a posttranslational modification characterized by the attachment of N-acetylglucosamine to specific serine or threonine residues. Unlike other protein glycosylations, O-GlcNAc modifications occur within the nucleus and cytoplasm. They are found on many cellular proteins, including nuclear pore, oncogene, cytoskeletal, heat shock, viral and transcription regulatory proteins. O-GlcNAc glycosylations are thought to obscure phosphorylation sites, counteracting phosphorylation-dependent signaling pathways and protein interactions.
For Research Use Only. Not for use in diagnostic procedures. Not for resale without express authorization.
How to use the Panel Builder
Watch the video to learn how to use the Invitrogen Flow Cytometry Panel Builder to build your next flow cytometry panel in 5 easy steps.
References (0)
Bioinformatics
Protein Aliases: GlcNAc

Performance Guarantee
If an Invitrogen™ antibody doesn't perform as described on our website or datasheet,we'll replace the product at no cost to you, or provide you with a credit for a future purchase.*
Learn more
We're here to help
Get expert recommendations for common problems or connect directly with an on staff expert for technical assistance related to applications, equipment and general product use.
Contact tech support