Search Thermo Fisher Scientific
Invitrogen
Phospho-ErbB2 (HER-2) (Tyr1248) Polyclonal Antibody
This Antibody was verified by Cell treatment to ensure that the antibody binds to the antigen stated.
FIGURE: 1 / 3
Phospho-ErbB2 (HER-2) (Tyr1248) Antibody (44904G) in WB
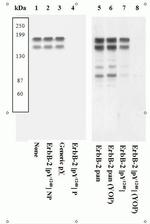


Product Details
44904G
Species Reactivity
Host/Isotype
Class
Type
Immunogen
Conjugate
Form
Purification
Storage buffer
Contains
Storage conditions
Shipping conditions
RRID
Target Information
ErbB2 (HER2) is a receptor tyrosine kinase that is overexpressed in some breast tumors. Herceptin, used in treatment of metastatic Her2-positive cancer, is monoclonal antibody targeting this kinase.
For Research Use Only. Not for use in diagnostic procedures. Not for resale without express authorization.
References (0)
Bioinformatics
Protein Aliases: c-erb B2/neu protein; CD antigen CD340; CD340; erb-b2; erbb 2; ErbB2 (pTyr1139); ErbB2 (pY1139); ErbB2 phospho Y1139; her 2; herstatin; human epidermal growth factor receptor 2; metas; Metastatic lymph node gene 19 protein; MLN 19; neuro/glioblastoma derived oncogene homolog; neuroblastoma/glioblastoma derived oncogene homolog; p185erbB2; phospho ErbB2; Proto-oncogene c-ErbB-2; Proto-oncogene Neu; Receptor tyrosine-protein kinase erbB-2; Tyrosine kinase-type cell surface receptor HER2; v-erb-b2 avian erythroblastic leukemia viral oncogene homolog 2; v-erb-b2 avian erythroblastic leukemia viral oncoprotein 2; v-erb-b2 erythroblastic leukemia viral oncogene homolog 2, neuro/glioblastoma derived oncogene homolog
Gene Aliases: CD340; ERBB2; HER-2; HER-2/neu; HER2; MLN 19; MLN19; NEU; NGL; TKR1
UniProt ID: (Human) P04626
Entrez Gene ID: (Human) 2064
Molecular Function:
![]() transmembrane signal receptor
transmembrane signal receptor

Performance Guarantee
If an Invitrogen™ antibody doesn't perform as described on our website or datasheet,we'll replace the product at no cost to you, or provide you with a credit for a future purchase.*
Learn more
We're here to help
Get expert recommendations for common problems or connect directly with an on staff expert for technical assistance related to applications, equipment and general product use.
Contact tech support
