Product References
Endocrine-Exocrine Signaling Drives Obesity-Associated Pancreatic Ductal Adenocarcinoma.
Cell
Chung KM,Singh J,Lawres L,Dorans KJ,Garcia C,Burkhardt DB,Robbins R,Bhutkar A,Cardone R,Zhao X,Babic A,Vayrynen SA,Dias Costa A,Nowak JA,Chang DT,Dunne RF,Hezel AF,Koong AC,Wilhelm JJ,Bellin MD,Nylander V,Gloyn AL,McCarthy MI,Kibbey RG,Krishnaswamy S,Wolpin BM,Jacks T,Fuchs CS,Muzumdar MD
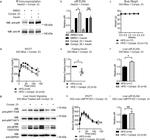
44-804G was used in Immunohistochemistry to argue that PDAC progression is driven by local obesity-associated changes in the tumor microenvironment and implicate endocrine-exocrine signaling beyond insulin in PDAC development.
Thu May 14 00:00:00 EDT 2020
IGF2 Autocrine-Mediated IGF1R Activation Is a Clinically Relevant Mechanism of Osimertinib Resistance in Lung Cancer.
Molecular cancer research : MCR
Manabe T,Yasuda H,Terai H,Kagiwada H,Hamamoto J,Ebisudani T,Kobayashi K,Masuzawa K,Ikemura S,Kawada I,Hayashi Y,Fukui K,Horimoto K,Fukunaga K,Soejima K
44-804G was used in Immunohistochemistry to find that IGF2 autocrine-mediated IGF1R pathway activation is a clinically relevant and common mechanism of acquired resistance to osimertinib.
Wed Apr 01 00:00:00 EDT 2020
The inhibitory effect of ECG and EGCG dimeric procyanidins on colorectal cancer cells growth is associated with their actions at lipid rafts and the inhibition of the epidermal growth factor receptor signaling.
Biochemical pharmacology
Zhu W,Li MC,Wang FR,Mackenzie GG,Oteiza PI
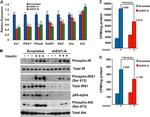
44-804G was used in Western Blotting to shed light on a potential molecular mechanism on how procyanidins-rich diets may lower CRC risk.
Fri May 01 00:00:00 EDT 2020
Lipid and glucose metabolism in hepatocyte cell lines and primary mouse hepatocytes: a comprehensive resource for in vitro studies of hepatic metabolism.
American journal of physiology. Endocrinology and metabolism
Nagarajan SR,Paul-Heng M,Krycer JR,Fazakerley DJ,Sharland AF,Hoy AJ
44-804G was used in Western Blotting to describe substrate metabolism of cultured AML12, IHH, and PH5CH8 cells, hepatocellular carcinoma-derived HepG2s, and primary mouse hepatocytes.
Mon Apr 01 00:00:00 EDT 2019
Ablation of Grb10 Specifically in Muscle Impacts Muscle Size and Glucose Metabolism in Mice.
Endocrinology
Holt LJ,Brandon AE,Small L,Suryana E,Preston E,Wilks D,Mokbel N,Coles CA,White JD,Turner N,Daly RJ,Cooney GJ
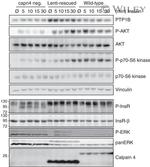
44-804G was used in Western Blotting to conclude that ablation of Grb10 in muscle is sufficient to affect muscle size and metabolism, supporting an important role for this protein in growth and metabolic pathways.
Thu Mar 01 00:00:00 EST 2018
Diabetes reversal by inhibition of the low-molecular-weight tyrosine phosphatase.
Nature chemical biology
Stanford SM,Aleshin AE,Zhang V,Ardecky RJ,Hedrick MP,Zou J,Ganji SR,Bliss MR,Yamamoto F,Bobkov AA,Kiselar J,Liu Y,Cadwell GW,Khare S,Yu J,Barquilla A,Chung TDY,Mustelin T,Schenk S,Bankston LA,Liddington RC,Pinkerton AB,Bottini N
Published figure using Phospho-IR/IGF1R (Tyr1162, Tyr1163) polyclonal antibody (Product # 44-804G) in Western Blot
Thu Jun 01 00:00:00 EDT 2017
Epigenetic regulation of macrophage polarization and inflammation by DNA methylation in obesity.
JCI insight
Wang X,Cao Q,Yu L,Shi H,Xue B,Shi H
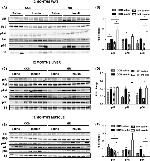
44804G was used in western blot to show that DNA hypermethylation at the PPARgamma1 promoter is induced by obesity-related factors
Thu Nov 17 00:00:00 EST 2016
Hepatocyte Nicotinamide Adenine Dinucleotide Phosphate Reduced Oxidase 4 Regulates Stress Signaling, Fibrosis, and Insulin Sensitivity During Development of Steatohepatitis in Mice.
Gastroenterology
Bettaieb A,Jiang JX,Sasaki Y,Chao TI,Kiss Z,Chen X,Tian J,Katsuyama M,Yabe-Nishimura C,Xi Y,Szyndralewiez C,Schröder K,Shah A,Brandes RP,Haj FG,Török NJ
44-804G was used in western blot to investigate the effects of nicotinamide adenine dinucleotide phosphate reduced oxidase 4 in liver tissues from patients with NASH and mice with steatohepatitis.
Sat Aug 01 00:00:00 EDT 2015
Agonistic aptamer to the insulin receptor leads to biased signaling and functional selectivity through allosteric modulation.
Nucleic acids research
Yunn NO,Koh A,Han S,Lim JH,Park S,Lee J,Kim E,Jang SK,Berggren PO,Ryu SH
44-804G was used in Western Blotting to investigate the potential of aptamers to the insulin receptor as target-specific agonists.
Fri Sep 18 00:00:00 EDT 2015
The insulin and IGF1 receptor kinase domains are functional dimers in the activated state.
Nature communications
Cabail MZ,Li S,Lemmon E,Bowen ME,Hubbard SR,Miller WT
Published figure using Phospho-IR/IGF1R (Tyr1162, Tyr1163) polyclonal antibody (Product # 44-804G) in Western Blot
Wed Mar 11 00:00:00 EDT 2015
Targeting the insulin-like growth factor receptor and Src signaling network for the treatment of non-small cell lung cancer.
Molecular cancer
Min HY,Yun HJ,Lee JS,Lee HJ,Cho J,Jang HJ,Park SH,Liu D,Oh SH,Lee JJ,Wistuba II,Lee HY
44804G was used in immunohistochemistry to study mechanisms that confer resistance to therapeutic interventions in the insulin-like growth factor receptor pathway
Thu Jun 04 00:00:00 EDT 2015
Methionine restriction restores a younger metabolic phenotype in adult mice with alterations in fibroblast growth factor 21.
Aging cell
Lees EK,Król E,Grant L,Shearer K,Wyse C,Moncur E,Bykowska AS,Mody N,Gettys TW,Delibegovic M
44-804G was used in western blot to examine the ability of methionine restriction to reverse age-induced obesity and insulin resistance in adult mice
Wed Oct 01 00:00:00 EDT 2014
Cardiac-specific adipose triglyceride lipase overexpression protects from cardiac steatosis and dilated cardiomyopathy following diet-induced obesity.
International journal of obesity (2005)
Pulinilkunnil T,Kienesberger PC,Nagendran J,Sharma N,Young ME,Dyck JR
44-804G was used in western blot to study the protective effects against cardiac steatosis and dilated cardiomyopathy of specifically overexpressing triglyceride lipase in cardiomyocytes in a murine model of diet-induced obesity.
Sat Feb 01 00:00:00 EST 2014
Fenretinide treatment prevents diet-induced obesity in association with major alterations in retinoid homeostatic gene expression in adipose, liver, and hypothalamus.
Diabetes
Mcilroy GD,Delibegovic M,Owen C,Stoney PN,Shearer KD,McCaffery PJ,Mody N
44-804G was used in western blot to test if alterations in retinoic acid-responsive genes contribute to the beneficial effects of fenretinide on obesity and insulin resistance.
Fri Mar 01 00:00:00 EST 2013
Early B-cell factor-1 (EBF1) is a key regulator of metabolic and inflammatory signaling pathways in mature adipocytes.
The Journal of biological chemistry
Griffin MJ,Zhou Y,Kang S,Zhang X,Mikkelsen TS,Rosen ED
Published figure using Phospho-IR/IGF1R (Tyr1162, Tyr1163) polyclonal antibody (Product # 44-804G) in Western Blot
Fri Dec 13 00:00:00 EST 2013
Reducing amyloid-related Alzheimer's disease pathogenesis by a small molecule targeting filamin A.
The Journal of neuroscience : the official journal of the Society for Neuroscience
Wang HY,Bakshi K,Frankfurt M,Stucky A,Goberdhan M,Shah SM,Burns LH
Wed Jul 18 00:00:00 EDT 2012
Calorie restriction enhances insulin-stimulated glucose uptake and Akt phosphorylation in both fast-twitch and slow-twitch skeletal muscle of 24-month-old rats.
The journals of gerontology. Series A, Biological sciences and medical sciences
Sequea DA,Sharma N,Arias EB,Cartee GD
44-804G was used in western blot to examine the insulin signaling pathway in isolated epitrochlearis and soleus muscles of old rats.
Sat Dec 01 00:00:00 EST 2012
Estrogen upregulates the IGF-1 signaling pathway in lung cancer through estrogen receptor-β.
Medical oncology (Northwood, London, England)
Tang H,Liao Y,Chen G,Xu L,Zhang C,Ju S,Zhou S
44-804G was used in immunohistochemistry - paraffin section to study estrogen regulated IGF-R signaling in non-small cell lung cancer.
Sat Dec 01 00:00:00 EST 2012
Conformation-sensing antibodies stabilize the oxidized form of PTP1B and inhibit its phosphatase activity.
Cell
Haque A,Andersen JN,Salmeen A,Barford D,Tonks NK
Fri Sep 30 00:00:00 EDT 2011
Knockdown of the Alström syndrome-associated gene Alms1 in 3T3-L1 preadipocytes impairs adipogenesis but has no effect on cell-autonomous insulin action.
International journal of obesity (2005)
Huang-Doran I,Semple RK
44-804G was used in western blot to knockdown Alms1 to investigate its role in adipocytes
Fri Oct 01 00:00:00 EDT 2010
BMS-536924 reverses IGF-IR-induced transformation of mammary epithelial cells and causes growth inhibition and polarization of MCF7 cells.
Clinical cancer research : an official journal of the American Association for Cancer Research
Litzenburger BC,Kim HJ,Kuiatse I,Carboni JM,Attar RM,Gottardis MM,Fairchild CR,Lee AV
Published figure using Phospho-IR/IGF1R (Tyr1162, Tyr1163) polyclonal antibody (Product # 44-804G) in Western Blot
Thu Jan 01 00:00:00 EST 2009
Calpain-mediated degradation of reversibly oxidized protein-tyrosine phosphatase 1B.
The FEBS journal
Trümpler A,Schlott B,Herrlich P,Greer PA,Böhmer FD
Published figure using Phospho-IR/IGF1R (Tyr1162, Tyr1163) polyclonal antibody (Product # 44-804G) in Western Blot
Thu Oct 01 00:00:00 EDT 2009
Constitutively active type I insulin-like growth factor receptor causes transformation and xenograft growth of immortalized mammary epithelial cells and is accompanied by an epithelial-to-mesenchymal transition mediated by NF-kappaB and snail.
Molecular and cellular biology
Kim HJ,Litzenburger BC,Cui X,Delgado DA,Grabiner BC,Lin X,Lewis MT,Gottardis MM,Wong TW,Attar RM,Carboni JM,Lee AV
44-804G was used in Western Blotting to examine the transforming potential of type I insulin-like growth factor receptor in human mammary epithelial cells.
Sun Apr 01 00:00:00 EDT 2007







![]() transmembrane signal receptor
transmembrane signal receptor



