Search
Thermo Scientific Chemicals
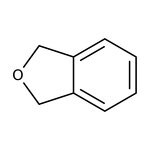
Ftalano, 98 %, Thermo Scientific Chemicals
CAS: 496-14-0 | C8H8O | 120.151 g/mol
Número de catálogo A10217.06
también denominado A10217-06
Precio (USD)
-
Cantidad:
5 g
Identificadores químicos
CAS496-14-0
IUPAC Name1,3-dihydro-2-benzofuran
Molecular FormulaC8H8O
InChI KeySFLGSKRGOWRGBR-UHFFFAOYSA-N
SMILESC1OCC2=CC=CC=C12
Ver más
Especificaciones Hoja de especificaciones
Hoja de especificaciones
Refractive Index1.5440-1.5480 @ 20?C
FormLiquid
Assay (GC)≥97.5%
Appearance (Color)Clear colorless to pale yellow
Phthalan is used in the synthesis of nine-membered cyclic dione ethers. It is used along with excess Li metal in the presence of 4,4'-Di-tert-butylbiphenyl, B21470, results in opening of the furan ring. It is used in the synthesis of functionalized benzylic alcohols.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Aplicaciones
El ftalano se utiliza en la síntesis de los éteres de dionas cíclicas de nueve miembros. Se utiliza junto con el exceso de metal de Li en presencia de 4,4′-Di-terc-butilbifenilo, B21470. Da como resultado la apertura del anillo de furano. Se utiliza en la síntesis de alcoholes benzílicos funcionales.
Solubilidad
Baja solubilidad acuosa.
Notas
Almacenar alejado de agentes oxidantes. Almacenar en un lugar fresco y seco y en recipientes bien sellados.
El ftalano se utiliza en la síntesis de los éteres de dionas cíclicas de nueve miembros. Se utiliza junto con el exceso de metal de Li en presencia de 4,4′-Di-terc-butilbifenilo, B21470. Da como resultado la apertura del anillo de furano. Se utiliza en la síntesis de alcoholes benzílicos funcionales.
Solubilidad
Baja solubilidad acuosa.
Notas
Almacenar alejado de agentes oxidantes. Almacenar en un lugar fresco y seco y en recipientes bien sellados.
RUO – Research Use Only
General References:
- Katsuhiko Tomooka; Lan-Fang Wang; Fumiaki Okazaki; Takeshi Nakai. External chiral ligand-induced enantioselective lithiation/SE2 reactions of isochroman and phthalan. Tetrahedron Letters. 2000, 41 6121-6125.
- Bondoc E.; Sakurai S.; Morris K.; Chiang WY.; Laane J. Fluorescence and electronic absorption spectra of phthalan: Two-dimensional vibrational potential energy surface for the ring-puckering and flapping in the S-1(pi,pi(*)) state. Journal of Chemical Physics. 2000, 112 (15), 6700-6706.
- For use in synthesis of nine-membered cyclic dione ethers, see: Synlett, 909 (1993).
- Treatment of phthalan with excess Li metal in the presence of 4,4'-Di-tert-butyl biphenyl , B21470, results in opening of the furan ring. Sequential introduction of two different electrophiles can be effected in the resulting bis-benzylic species, consequently phthalan can be regarded as an o-xylene dianion synthon. This approach provides a convenient access to diols and hydroxy acids which can be readily dehydrated to benzo-fused 6- or 7-membered cyclic ethers or lactones: Tetrahedron, 51, 3351 (1995); see also: J. Org. Chem., 61, 4913 (1996); Tetrahedron Lett., 39, 7759 (1998).