Search
Thermo Scientific Chemicals
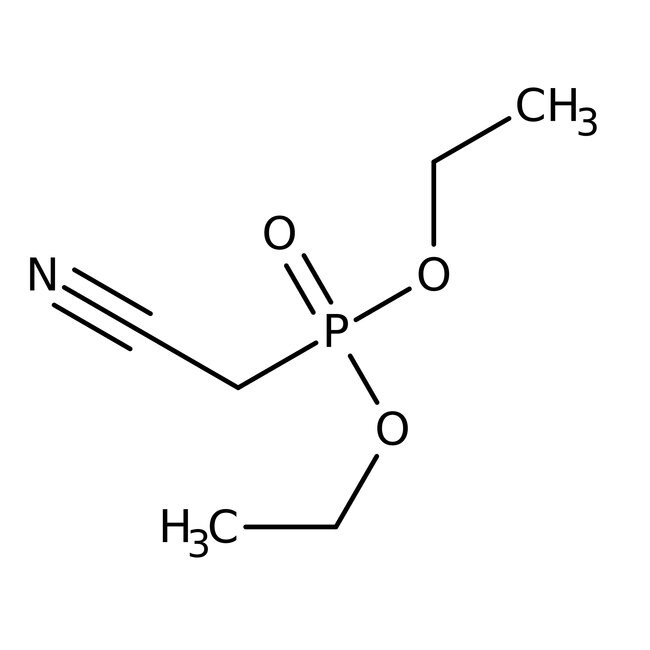
Diethyl cyanomethylphosphonate, 96%
CAS: 2537-48-6 | C6H12NO3P | 177.14 g/mol
Catalog number A10218.14
also known as A10218-14
Price (EUR)
83,00
Each
Quantity:
25 g
Price (EUR)
83,00
Each
Chemical Identifiers
CAS2537-48-6
IUPAC Namediethyl (cyanomethyl)phosphonate
Molecular FormulaC6H12NO3P
InChI KeyKWMBADTWRIGGGG-UHFFFAOYSA-N
SMILESCCOP(=O)(CC#N)OCC
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Clear colorless to pale yellow
FormLiquid
Assay (GC)≥95.0%
Refractive Index1.4290-1.4350 @ 20?C
Diethyl cyanomethyl phosphonate is used as an intermediate in Horner-Emmons reaction for the synthesis of substituted nitriles and their amide and heterocyclic derivatives. It is known as a modified Wittig reagent used in the preparation of alpha, beta-unsaturated nitriles from ketones or aldehydes like 3-hydroxy-3-methylbutanal. It reacts with epoxides and nitrones to prepare cyano-substituted cyclopropanes and aziridines respectively. It is actively involved in the synthesis of alpha-arylated alkanenitriles via reaction with aryl iodides in presence of CuI.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Diethyl cyanomethyl phosphonate is used as an intermediate in Horner-Emmons reaction for the synthesis of substituted nitriles and their amide and heterocyclic derivatives. It is known as a modified Wittig reagent used in the preparation of alpha, beta-unsaturated nitriles from ketones or aldehydes like 3-hydroxy-3-methylbutanal. It reacts with epoxides and nitrones to prepare cyano-substituted cyclopropanes and aziridines respectively. It is actively involved in the synthesis of alpha-arylated alkanenitriles via reaction with aryl iodides in presence of CuI.
Solubility
Miscible with water, chloroform, terahydrofuran, ethyl acetate and methylene chloride.
Notes
Incompatible with acids, bases, oxidizing agents and reducing agents.
Diethyl cyanomethyl phosphonate is used as an intermediate in Horner-Emmons reaction for the synthesis of substituted nitriles and their amide and heterocyclic derivatives. It is known as a modified Wittig reagent used in the preparation of alpha, beta-unsaturated nitriles from ketones or aldehydes like 3-hydroxy-3-methylbutanal. It reacts with epoxides and nitrones to prepare cyano-substituted cyclopropanes and aziridines respectively. It is actively involved in the synthesis of alpha-arylated alkanenitriles via reaction with aryl iodides in presence of CuI.
Solubility
Miscible with water, chloroform, terahydrofuran, ethyl acetate and methylene chloride.
Notes
Incompatible with acids, bases, oxidizing agents and reducing agents.
RUO – Research Use Only
General References:
- Horner-Wadsworth-Emmons olefination converts carbonyl compounds to substituted acrylonitriles: J. Am. Chem. Soc., 83, 1733 (1961); J. Org. Chem., 30, 505 (1965). LiOH has been found to be an effective base for this reaction: Tetrahedron Lett., 44, 1333 (2003). See Appendix 1. See also 3-Coumaranone, A10202.
- The active methylene group can be monoalkylated under phase transfer conditions with alkyl iodides or allylic or benzylic bromides: Synthesis, 516 (1975). Use of alumina as base leads predominantly to the Knoevenagel product rather than the Wadsworth-Emmons: Tetrahedron, 41, 1259 (1985).
- Grun, A.; Balint, E.; Keglevich, G. Solid-Liquid Phase C-Alkylation of Active Methylene Containing Compounds under Microwave Conditions. Catalysts 2015, 5 (2), 634-652.
- Dai, Y.; Yu, B. Total synthesis of astrosterioside A, an anti-inflammatory asterosaponin. Chem. Commun. 2015, 51 (72), 13826-13829.