Search
Thermo Scientific Chemicals
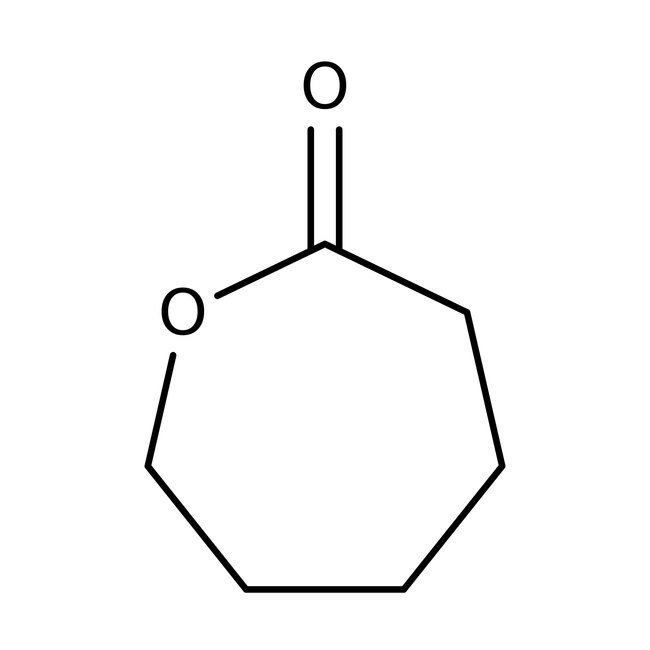
epsilon-Caprolactone, 99%
CAS: 502-44-3 | C6H10O2 | 114.144 g/mol
Catalog number A10299.36
also known as A10299-36
Price (TWD)
-
Quantity:
500 g
Chemical Identifiers
CAS502-44-3
IUPAC Nameoxepan-2-one
Molecular FormulaC6H10O2
InChI KeyPAPBSGBWRJIAAV-UHFFFAOYSA-N
SMILESO=C1CCCCCO1
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Clear colorless
Assay (GC)≥98.5%
Refractive Index1.4620-1.4650 @ 20?C
FormLiquid
Identification (FTIR)Conforms
ε-Caprolactone is widely used as a monomer in the manufacturing of highly specialized polymers. It is mainly utilized as a precursor to caprolactam. It finds an application in the synthesis of polyglecaprone, which is used as a suture material in surgery.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
ε-Caprolactone is widely used as a monomer in the manufacturing of highly specialized polymers. It is mainly utilized as a precursor to caprolactam. It finds an application in the synthesis of polyglecaprone, which is used as a suture material in surgery.
Solubility
Miscible with water and most organic solvents.
Notes
Keep the container tightly closed in a dry and well-ventilated place. Combustible liquid.
ε-Caprolactone is widely used as a monomer in the manufacturing of highly specialized polymers. It is mainly utilized as a precursor to caprolactam. It finds an application in the synthesis of polyglecaprone, which is used as a suture material in surgery.
Solubility
Miscible with water and most organic solvents.
Notes
Keep the container tightly closed in a dry and well-ventilated place. Combustible liquid.
RUO – Research Use Only
General References:
- Schmidt, S.; Scherkus, C.; Muschiol, J.; Menyes, U.; Winkler, T.; Hummel, W.; Gröger, H.; Liese, A.; Herz, H. G.; Bornscheuer, U. T. An Enzyme Cascade Synthesis of ε-Caprolactone and its Oligomers. Angew. Chem. Int. Ed. 2015, 54 (9), 2784-2787.
- Mrówczyński, W.; Mugnai, D.; Valence, S.; Tille, J. C.; Khabiri, E.; Cikirikcioglu, M.; Möller, M.; Walpoth, B. H. Porcine carotid artery replacement with biodegradable electrospun poly-e-caprolactone vascular prosthesis. J. Vasc. Surg. 2014, 59 (1), 210-219.
- Jones, G. O.; Chang, Y. A.; Horn, H. W.; Acharya, A. K.; Rice, J. E.; Hedrick, J. L.; Waymouth, R. M. N-Heterocyclic Carbene-Catalyzed Ring Opening Polymerization of ε-Caprolactone with and without Alcohol Initiators: Insights from Theory and Experiment. J. Phys. Chem. B 2015, 119 (17), 5728-5737.