Search
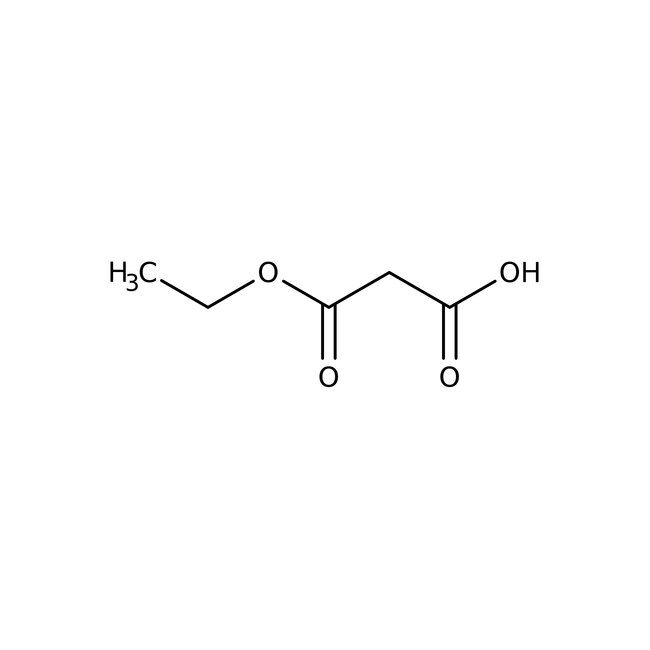
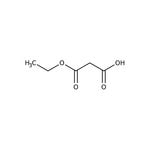
Ethyl potassium malonate, 98%
Ethyl potassium malonate is used as a competitive inhibitor of the enzyme succinate dehydrogenase. It acts as a precursor to produce (trimethylsilyl)ethyl malonate, which is utilized to prepare beta-ketoesters by acylation. Further, it reacts with aryl nitriles to prepare beta-amino acrylates in the presence of zinc chloride and a catalytic amount of Hünig's base. In addition to this, it serves as an intermediate for the preparation of ethyl tert-butyl malonate.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Ethyl potassium malonate is used as a competitive inhibitor of the enzyme succinate dehydrogenase. It acts as a precursor to produce (trimethylsilyl)ethyl malonate, which is utilized to prepare beta-ketoesters by acylation. Further, it reacts with aryl nitriles to prepare beta-amino acrylates in the presence of zinc chloride and a catalytic amount of Hünig′s base. In addition to this, it serves as an intermediate for the preparation of ethyl tert-butyl malonate.
Solubility
Soluble in water.
Notes
Hygroscopic. Incompatible with strong oxidizing agents.
General References:
- An improved method for acylation with acid chlorides utilizes the Rathke MgCl2-triethylamine reagent in acetonitrile or ethyl acetate to give ß-keto esters in very high yields: Synthesis, 290 (1993):
- Compare Diethyl malonate, A15468 and Dimethyl malonate, A11007, for other applications of this technique. See also Ethyl hydrogen malonate, A12627.
- Alternatively, ß-keto esters can be obtained in high yield by reaction with TMS chloride to generate ethyl trimethylsilyl malonate, followed by in situ reaction with acyl imidazoles or acyl chlorides in the presence of DBU: Tetrahedron Lett., 35, 9323 (1994).
- Tancock, J.; Wirth, T. Selenium-Mediated Synthesis of Tetrasubstituted Naphthalenes through Rearrangement. Molecules 2015, 20 (6), 10866-10872.
- Stasyuk, A. J.; Smoleń, S.; Glodkowska-Mrowka, E.; Brutkowski, W.; Cyrański, M. K.; Tkachenko, N.; Gryko, D. T. Synthesis of Fluorescent Naphthoquinolizines via Intramolecular Houben-Hoesch Reaction. Chem. Asian J. 2015, 10 (3), 553-558.