Search
Thermo Scientific Chemicals
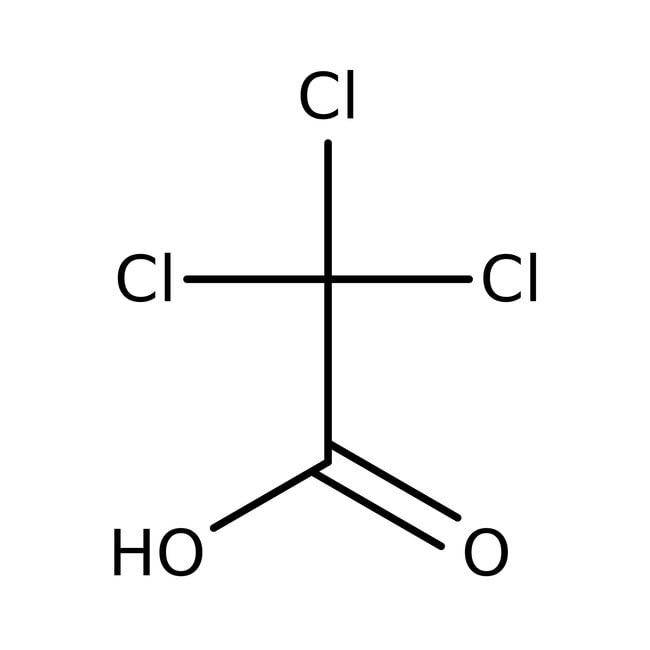
Ácido tricloroacético, 99 %, Thermo Scientific Chemicals
CAS: 76-03-9 | C2HCl3O2 | 163.378 g/mol
Número de catálogo A11156.30
Precio (USD)
-
Cantidad:
250g
Identificadores químicos
CAS76-03-9
IUPAC Nametrichloroacetic acid
Molecular FormulaC2HCl3O2
InChI KeyYNJBWRMUSHSURL-UHFFFAOYSA-N
SMILESOC(=O)C(Cl)(Cl)Cl
Ver más
Especificaciones Hoja de especificaciones
Hoja de especificaciones
Identification (FTIR)Conforms
Assay (Aqueous acid-base Titration)≥98.5 to ≤101.5%
Water Content (Karl Fischer Titration)<0.5%
Appearance (Color)White to pale yellow
Melting Point (clear melt)52-60?C
Ver más
Protein precipitation reagent Trichloroacetic acid is used as a precipitating agent in biochemistry for precipitation of proteins, DNA and RNA. It is an active ingredient used for cosmetic treatments like chemical peels, tattoo removal and the treatment of warts including genital warts. It is also used to determine protein concentration and a decalcifier and fixative in microscopy.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Aplicaciones
Reactivo de precipitación de proteínas El ácido tricloroacético se utiliza como agente precipitante en bioquímica para la precipitación de proteínas, ADN y ARN. Es un ingrediente activo utilizado para tratamientos cosméticos como exfoliación química, eliminación de tatuajes y el tratamiento de verrugas, incluyendo verrugas genitales. También se utiliza para determinar la concentración de proteínas y como descalcificador y fijador en microscopía.
Solubilidad
Soluble en agua, alcohol, éter etílico, benceno, acetona, o-xileno y tetracloruro de carbono.
Notas
Sensible a la humedad. Higroscópico. Almacenar en un lugar fresco. Incompatible con agentes oxidantes fuertes, bases fuertes y aminas.
Reactivo de precipitación de proteínas El ácido tricloroacético se utiliza como agente precipitante en bioquímica para la precipitación de proteínas, ADN y ARN. Es un ingrediente activo utilizado para tratamientos cosméticos como exfoliación química, eliminación de tatuajes y el tratamiento de verrugas, incluyendo verrugas genitales. También se utiliza para determinar la concentración de proteínas y como descalcificador y fijador en microscopía.
Solubilidad
Soluble en agua, alcohol, éter etílico, benceno, acetona, o-xileno y tetracloruro de carbono.
Notas
Sensible a la humedad. Higroscópico. Almacenar en un lugar fresco. Incompatible con agentes oxidantes fuertes, bases fuertes y aminas.
RUO – Research Use Only
General References:
- Trihaloacetic acids react with aldehydes in DMSO at room temperature to give trihalomethyl carbinols: J. Chem. Soc., Perkin 2, 1247 (1984):
- The acid and its Na salt (1:1) in DMF are also effective: Tetrahedron Lett., 33, 3435 (1992). Use of HMPA or DMI extends the scope to less reactive aldehydes: Synthesis, 327 (1990).
- Malachová, A.; Štočková, L.; Wakker, A.; Varga, E.; Krska, R.; Michlmayr, H.; Adam, G.; Berthiller, F. Critical evaluation of indirect methods for the determination of deoxynivalenol and its conjugated forms in cereals. Anal. Bioanal. Chem. 2015, 407 (20), 6009-6020.
- Young, M. S.; van Tran, K.; Goh, E.; Shia, J. C. A Rapid SPE-Based Analytical Method for UPLC/MS/MS Determination of Aminoglycoside Antibiotic Residues in Bovine Milk, Muscle, and Kidney. J. AOAC Int. 2014, 97 (6), 1737-1741.