Search
Thermo Scientific Chemicals
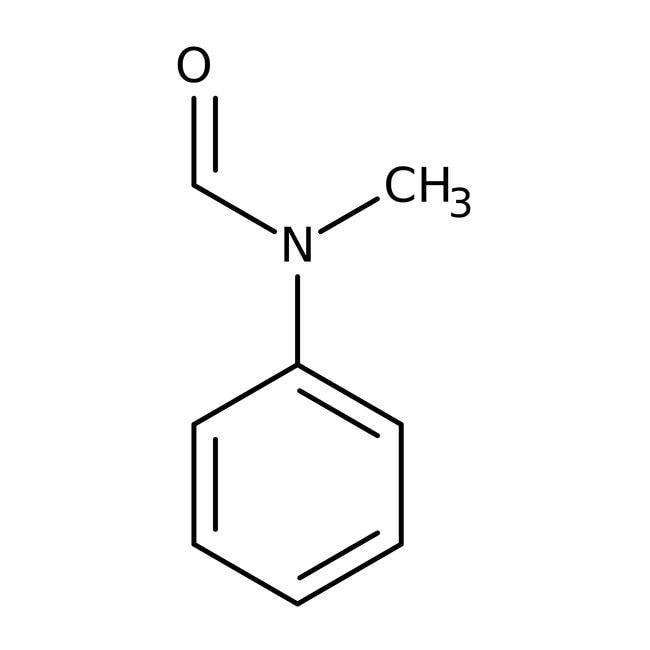
N-Methylformanilide, 99%
CAS: 93-61-8 | C8H9NO | 135.166 g/mol
Catalog number A11829.30
also known as A11829-30
Price (TWD)
-
Quantity:
250 g
Chemical Identifiers
CAS93-61-8
IUPAC NameN-methyl-N-phenylformamide
Molecular FormulaC8H9NO
InChI KeyJIKUXBYRTXDNIY-UHFFFAOYSA-N
SMILESCN(C=O)C1=CC=CC=C1
View more
Specifications Specification Sheet
Specification Sheet
Assay (GC)≥98.5%
Appearance (Color)Clear colorless to pale yellow
FormLiquid
Identification (FTIR)Conforms
Refractive Index1.5590-1.5630 @ 20?C
N-Methylformanilide is used as a formylating reagent for certain organometallics. It is in combination with phosphoryl chloride used for Vilsmeier-Haack reactions and in heterocycle syntheses. Further, it acts as a swelling agent in the dyeing process of meta-aramid fibers.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
N-Methylformanilide is used as a formylating reagent for certain organometallics. It is in combination with phosphoryl chloride used for Vilsmeier-Haack reactions and in heterocycle syntheses. Further, it acts as a swelling agent in the dyeing process of meta-aramid fibers.
Solubility
Immisible with water.
Notes
Incompatible with oxidizing agents.
N-Methylformanilide is used as a formylating reagent for certain organometallics. It is in combination with phosphoryl chloride used for Vilsmeier-Haack reactions and in heterocycle syntheses. Further, it acts as a swelling agent in the dyeing process of meta-aramid fibers.
Solubility
Immisible with water.
Notes
Incompatible with oxidizing agents.
RUO – Research Use Only
General References:
- Component of Vilsmeier's original reagent for the formylation of reactive aromatics via the iminium salts: Ber., 60, 119 (1927); much used in earlier work, where N,N-Dimethyl formamide, A13547 , is often now preferred due to lower cost, greater thermal stability of the iminium salt and easier separation of the by-products (water-soluble in the case of DMF). The N-methylformanilide reagent is still occasionally preferred because of its higher electrophilic potential, although this may be partially offset by its greater steric demand. For examples of its use, see: Org. Synth. Coll., 3, 96 (1955); 4, 915 (1963). Review: Adv. Org. Chem., 9, 225 (1976). Monograph: Synthesis Using Vilsmeier Reagents, C. M. Marson, P. R. Giles, CRC Press, Boca Raton, FL (1994).
- Has also been used for the conversion of aryllithiums to aldehydes: J. Chem. Soc. (C), 1700 (1969).
- Islam, M. T.; Aimone, F.; Ferri, A.; Rovero, G. Use of N-methylformanilide as swelling agent for meta-aramid fibers dyeing: Kinetics and equilibrium adsorption of Basic Blue 41. Dyes Pigm. 2015, 113, 554-561.
- Zhang, J.; Wang, Y.; Luo, N.; Chen, Z.; Wu, K.; Yin, G. Redox inactive metal ion triggered N-dealkylation by an iron catalyst with dioxygen activation: a lesson from lipoxygenases. Dalton Trans. 2015, 44 (21), 9847-9859.