Search
Thermo Scientific Chemicals
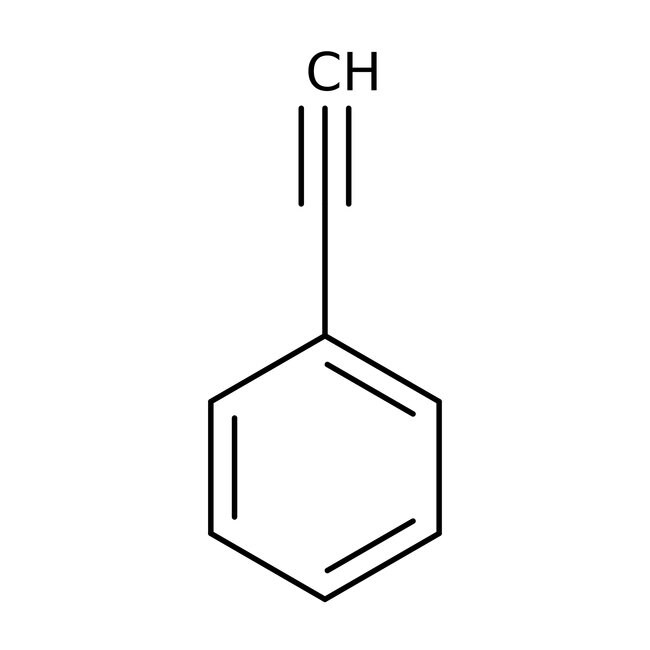
Phenylacetylene, 98+%
CAS: 536-74-3 | C8H6 | 102.136 g/mol
Catalog number A12139.14
also known as A12139-14
Price (EUR)
36,20
Each
Quantity:
25 g
Price (EUR)
36,20
Each
Chemical Identifiers
CAS536-74-3
IUPAC Nameethynylbenzene
Molecular FormulaC8H6
InChI KeyUEXCJVNBTNXOEH-UHFFFAOYSA-N
SMILESC#CC1=CC=CC=C1
View more
Specifications Specification Sheet
Specification Sheet
FormLiquid
Refractive Index1.5460-1.5500 @ 20?C
Assay (GC)≥98.0%
Appearance (Color)Clear colorless to yellow
Identification (FTIR)Conforms
Phenylacetylene is involved in the preparation of styrene by reduction using Lindlar catalyst. It is used to study the mechanism of the palladium-catalyzed phenyl acetylene oxidative carbonylation reaction. It is also used in the polymerization process to prepare polyphenylacetylene namely 1,2,4-triphenylbenzene and 1,3,5-triphenylbenzene.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Phenylacetylene is involved in the preparation of styrene by reduction using Lindlar catalyst. It is used to study the mechanism of the palladium-catalyzed phenyl acetylene oxidative carbonylation reaction. It is also used in the polymerization process to prepare polyphenylacetylene namely 1,2,4-triphenylbenzene and 1,3,5-triphenylbenzene.
Solubility
Immiscible with water.
Notes
Store in cool place. Moisture sensitive. Keep the container tightly closed in a dry and well-ventilated place. Incompatible with acids, halogens, alkali metals and oxidizing agents.
Phenylacetylene is involved in the preparation of styrene by reduction using Lindlar catalyst. It is used to study the mechanism of the palladium-catalyzed phenyl acetylene oxidative carbonylation reaction. It is also used in the polymerization process to prepare polyphenylacetylene namely 1,2,4-triphenylbenzene and 1,3,5-triphenylbenzene.
Solubility
Immiscible with water.
Notes
Store in cool place. Moisture sensitive. Keep the container tightly closed in a dry and well-ventilated place. Incompatible with acids, halogens, alkali metals and oxidizing agents.
RUO – Research Use Only
General References:
- Conversion to the 1-bromoalkyne has been described: Org. Synth. Coll., 5, 921 (1973). For an improved phase-transfer method, using NaBraOCl, see: J. Org. Chem., 57, 4555 (1992).
- For cyanation with CuCN in the presence of TMS chloride and NaI, see: Tetrahedron Lett., 34, 591 (1993).
- Glaser oxidative coupling by Cu(OAc)2 in pyridine gives diphenylbuta-1,3-diyne: Org. Synth. Coll., 5, 499 (1973).
- For conversion to the Grignard with ethylmagnesium bromide, and subsequent Cu-catalyzed coupling with allyl bromide, see: Org. Synth. Coll., 6, 925 (1988).
- For preparation of the cuprate and references for coupling with aryl halides to give diarylacetylenes, see: Org. Synth. Coll., 6, 916 (1988).
- Dimetallation can be accomplished with a 2:1 molar mixture of n-BuLi and KO-t-Bu in THF, the second metallation occurring specifically at the ortho-position. Thus, the bis(TMS)-derivative can be prepared in excellent yield: J. Org. Chem., 55, 1311 (1990).
- Weerachawanasak, P.; Praserthdam, P.; Panpranot, J. Liquid-Phase Hydrogenation of Phenylacetylene Over the Nano-Sized Pd/TiO2 Catalysts. J. Nanosci. Nanotechnol. 2014, 14 (4), 3170-3175.
- Yoshida, H.; Zama, T.; Fujita, S. I.; Panpranot, J.; Arai, M. Liquid phase hydrogenation of phenylacetylene over Pd and PdZn catalysts in toluene: effects of alloying and CO2 pressurization. RSC Adv. 2014, 4 (47), 24922-24928.