Search
Thermo Scientific Chemicals
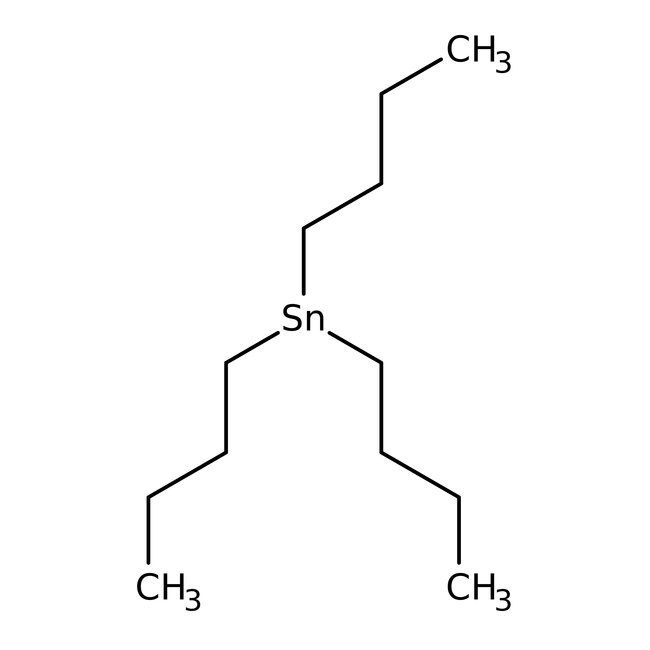
Tri-n-butyltin hydride, 97%
CAS: 688-73-3 | C12H27Sn | 290.058 g/mol
Catalog number A13298.22
also known as A13298-22
Price (USD)
-
Quantity:
100 g
Chemical Identifiers
CAS688-73-3
IUPAC Nametributylstannyl
Molecular FormulaC12H27Sn
InChI KeyPIILXFBHQILWPS-UHFFFAOYSA-N
SMILESCCCC[Sn](CCCC)CCCC
View more
Specifications Specification Sheet
Specification Sheet
FormLiquid
Refractive Index1.4705-1.4755 @ 20?C
CommentMay form slight precipitate on storage. May develop pressure.
Appearance (Color)Clear or slightly hazy, colorless
Assay (GC)≥96.0%
View more
Tri-n-butyltin hydride is used as a radical reagent in reductive cleavage, radical dehalogenation and intramolecular radical cyclization. It also promotes intramolecular cyclization leading to isoxazolo-benzaulene ring system. Further, it acts as a reducing agent for the conversion of alkyl halides to hydrocarbons. In addition to this, it is used in a veterinary anthelmintic for poultry and an intermediate to make other butyltin compounds.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Tri-n-butyltin hydride is used as a radical reagent in reductive cleavage, radical dehalogenation and intramolecular radical cyclization. It also promotes intramolecular cyclization leading to isoxazolo-benzaulene ring system. Further, it acts as a reducing agent for the conversion of alkyl halides to hydrocarbons. In addition to this, it is used in a veterinary anthelmintic for poultry and an intermediate to make other butyltin compounds.
Solubility
Immiscible with water.
Notes
Air and moisture sensitive. Store in a cool place. Incompatible with strong oxidizing agents and acids.
Tri-n-butyltin hydride is used as a radical reagent in reductive cleavage, radical dehalogenation and intramolecular radical cyclization. It also promotes intramolecular cyclization leading to isoxazolo-benzaulene ring system. Further, it acts as a reducing agent for the conversion of alkyl halides to hydrocarbons. In addition to this, it is used in a veterinary anthelmintic for poultry and an intermediate to make other butyltin compounds.
Solubility
Immiscible with water.
Notes
Air and moisture sensitive. Store in a cool place. Incompatible with strong oxidizing agents and acids.
RUO – Research Use Only
General References:
- Reducing agent and source of tributyltin radicals. Widely used for selective reduction of alkyl halides to alkanes by a radical chain mechanism. The reaction is tolerant of a wide range of functionality, including OH and NH, in contrast to polar metal hydride reagents. For reviews, see: Synthesis, 499 (1970); 665 (1987). For a brief feature on uses of this reagent in synthesis, see: Synlett, 173 (2007). For use in the synthesis of deoxy sugars via reductive rearrangement of glycosyl bromides, see: Org. Synth. Coll., 8, 583 (1993). Cyclization of the initially-formed alkyl radical to a suitably positioned double bond may occur. Formation of 5-membered rings is strongly favoured, and ring closure onto an existing ring gives the cis-fused product. See, e.g.: J. Am. Chem. Soc., 108, 5893 (1986):
- Intermolecular coupling with electron-deficient alkenes is also effective: Angew. Chem. Int. Ed., 23, 69 (1984); Org. Synth. Coll., 8, 148 (1993). Review: Synthesis, 417 (1988).
- Secondary alcohols can be deoxygenated by reduction of their xanthate derivatives: J. Chem. Soc., Perkin 1, 1574 (1975); Org. Synth. Coll., 7, 139 (1990), avoiding the rearrangements encountered with carbocation-based methods.
- TBTH also cleaves other C-heteroatom bonds, controlled by the stability of the resulting radical: Nitro-groups at tertiary centers are readily cleaved: Synthesis, 693 (1986). Azides are readily reduced to amines: Synlett, 342 (1991). C-S and C-Se bonds are also cleaved. J. Am. Chem. Soc., 104, 2046 (1982); 112, 4008 (1990); J. Org. Chem., 49, 5206 (1984); 54, 1234 (1989).
- Dialdehydes and keto aldehydes undergo free-radical intramolecular pinacol coupling to give cyclic diols: J. Am. Chem. Soc., 117, 7283 (1995); J. Org. Chem., 63, 6357 (1998).