Search
Thermo Scientific Chemicals
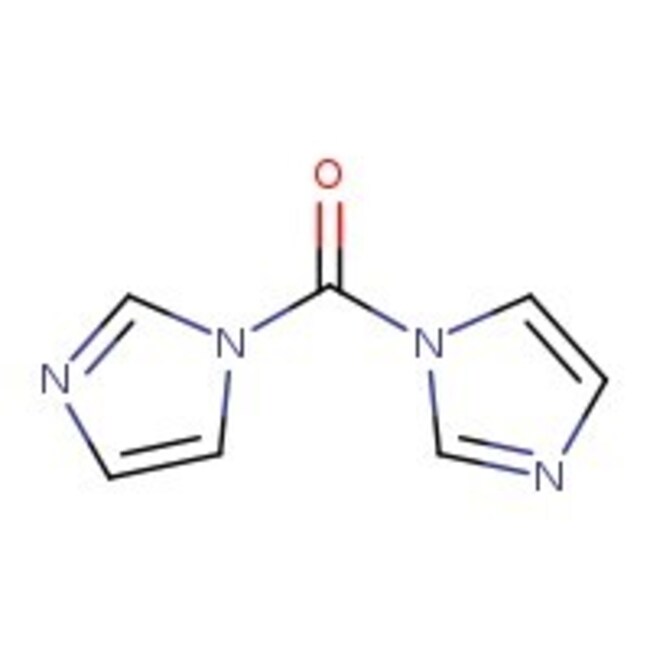
1,1'-Carbonildiimidazol, 97 %, Thermo Scientific Chemicals
CAS: 530-62-1 | C7H6N4O | 162.15 g/mol
Número de catálogo A14688.18
Precio (USD)
-
Cantidad:
50 g
Identificadores químicos
CAS530-62-1
IUPAC Name1-(1H-imidazole-1-carbonyl)-1H-imidazole
Molecular FormulaC7H6N4O
InChI KeyPFKFTWBEEFSNDU-UHFFFAOYSA-N
SMILESO=C(N1C=CN=C1)N1C=CN=C1
Ver más
Especificaciones Hoja de especificaciones
Hoja de especificaciones
FormCrystals or powder or crystalline powder
Assay (Non-aqueous acid-base Titration)≥96.0 to ≤104.0% (total imidazole content)
Solution Test5% solution in DMF is clear
Appearance (Color)White to cream
Identification (FTIR)Conforms
Peptide coupling reagent1,1'-Carbonyldiimidazole acts as a coupling reagent and utilized for coupling of amino acids in order to prepare peptide in organic synthesis. It is also used in the preparation of beta-keto sulfones, sulfoxides and beta-enamino acid derivatives. It is used to convert alcohols and amines into carbamates, esters, and ureas. It is involved in the preparation of formylized imidazole by reaction with formic acid. Further, it is used in the synthesis of dipolar polyamides compounds. In addition to this, it is considered as an equivalent of phosgene and used to prepare asymmetric bis alkyl carbonate.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Aplicaciones
Reactivo de acoplamiento de péptidos. El 1,1′-carbonildiimidazol actúa como reactivo de acoplamiento y se utiliza para el acoplamiento de amino ácidos con el fin de preparar péptido en síntesis orgánica. También se utiliza en la preparación de derivados de ácido de beta-enamina, beta-cetosulfonas y sulfóxidos. Se utiliza para convertir alcoholes y aminas en carbamatos, ésteres y ureas. Participa en la preparación de imidazol formilado por reacción con ácido fórmico. Además, se utiliza en la síntesis de compuestos de poliamidas dipolares. Aparte de esto, se considera un equivalente de fosgeno y se utiliza para preparar carbonato de bisalquilo asimétrico.
Solubilidad
Soluble en dimetilformamida.
Notas
Almacenar en un lugar fresco. Incompatible con agua, ácidos fuertes, agentes oxidantes fuertes, bases fuertes y aminas.
Reactivo de acoplamiento de péptidos. El 1,1′-carbonildiimidazol actúa como reactivo de acoplamiento y se utiliza para el acoplamiento de amino ácidos con el fin de preparar péptido en síntesis orgánica. También se utiliza en la preparación de derivados de ácido de beta-enamina, beta-cetosulfonas y sulfóxidos. Se utiliza para convertir alcoholes y aminas en carbamatos, ésteres y ureas. Participa en la preparación de imidazol formilado por reacción con ácido fórmico. Además, se utiliza en la síntesis de compuestos de poliamidas dipolares. Aparte de esto, se considera un equivalente de fosgeno y se utiliza para preparar carbonato de bisalquilo asimétrico.
Solubilidad
Soluble en dimetilformamida.
Notas
Almacenar en un lugar fresco. Incompatible con agua, ácidos fuertes, agentes oxidantes fuertes, bases fuertes y aminas.
RUO – Research Use Only
General References:
- Reagent for peptide coupling via the acylimidazolide: Liebigs Ann. Chem., 609, 75 (1957); J. Am. Chem. Soc., 80, 4423 (1958); 82, 4596 (1960): J. Org. Chem., 27, 2094 (1962). For peptide reagents, see Appendix 6.
- One-pot esterification of a carboxylic acid with t-BuOH occurs in the presence of DBU: Synthesis, 833 (1982). The reactivity of acyl imidazolides can be increased by N-alkylation: Chem. Pharm. Bull., 32, 5044 (1984).
- Acylimidazolides can also be reduced to aldehydes by DIBAL-H. This reaction has been applied to N-protected amino acids: J. Chem. Soc., Chem. Commun., 79 (1979).
- Dehydrates aldoximes, including chiral oximes, to nitriles in high yield: J. Chem. Soc., Chem. Commun., 628 (1973); Synth. Commun., 12, 25 (1982). Ketoximes can be converted to amides by the spontaneous Beckmann rearrangement of imidazolium salts: Chem. Pharm. Bull., 32, 2560 (1984):
- ß-Hydroxy amino acids are dehydrated to dehydroamino acids: Synthesis, 968 (1982).
- ɑß-Dihydroxyketones are converted toɑ-diketones: Synth. Commun., 23, 2219 (1993).
- Lanzillotto, M.; Konnert, L.; Lamaty, F.; Martinez, J.; Colacino, E. Mechanochemical 1,1'-Carbonyldiimidazole-Mediated Synthesis of Carbamates. ACS Sustainable Chem. Eng. 2015, 3 (11), 2882-2889.
- Rodrigues, M. T.; Santos, M. S.; Santos, H.; Coelho, F. 1, 1'-Carbonyldiimidazole mediates the synthesis of N-substituted imidazole derivatives from Morita-Baylis-Hillman adducts. Tetrahedron Lett. 2014, 55 (1), 180-183.
- Lafrance, D.; Bowles, P.; Leeman, K.; Rafka, R. Mild Decarboxylative Activation of Malonic Acid Derivatives by 1, 1'-Carbonyldiimidazole. Org. Lett. 2011, 13 (9), 2322-2325.