Search
Thermo Scientific Chemicals
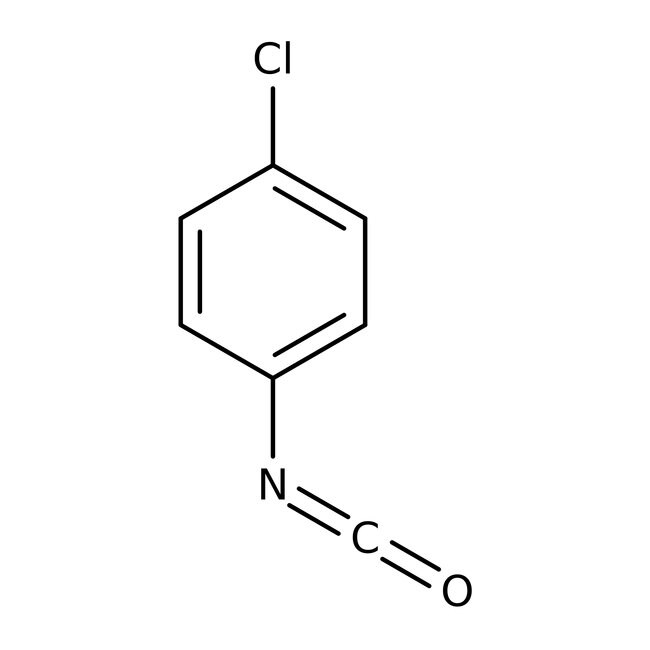
4-Clorofenilisocianato, 98 %, Thermo Scientific Chemicals
CAS: 104-12-1 | C7H4ClNO | 153.565 g/mol
Número de catálogo A14966.22
también denominado A14966-22
Precio (USD)
-
Cantidad:
100 g
Identificadores químicos
CAS104-12-1
IUPAC Name1-chloro-4-isocyanatobenzene
Molecular FormulaC7H4ClNO
InChI KeyADAKRBAJFHTIEW-UHFFFAOYSA-N
SMILESClC1=CC=C(C=C1)N=C=O
Ver más
Especificaciones Hoja de especificaciones
Hoja de especificaciones
CommentMay develop some turbidity or precipitate
Melting Point29-33?C
Assay (GC)≥97.5%
Appearance (Color)White to cream to yellow
FormCrystalline or fused solid
4-Chlorophenyl isocyanate is used in the preparation of isothiocyanates. Aryl isocyanates undergo [2+2] cycloaddition reactions with dihydrofuran to give bicyclic β-lactams in high yield.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Aplicaciones
El 4-clorofenilisocianato se utiliza en la preparación de isotiocianatos. Los isotiocianatosde arilo se someten a reacciones de cicloadición [2+2] con dihidrofurano para producir betalactámicos bicíclicos de alto rendimiento.
Solubilidad
Hidroliza en agua.
Notas
Mantener en un recipiente herméticamente cerrado. Almacenar lejos de agentes oxidantes.
El 4-clorofenilisocianato se utiliza en la preparación de isotiocianatos. Los isotiocianatosde arilo se someten a reacciones de cicloadición [2+2] con dihidrofurano para producir betalactámicos bicíclicos de alto rendimiento.
Solubilidad
Hidroliza en agua.
Notas
Mantener en un recipiente herméticamente cerrado. Almacenar lejos de agentes oxidantes.
RUO – Research Use Only
General References:
- Robert L. Metcalf.; Po-Yung. Lu.; Stephen. Bowlus. Degradation and environmental fate of 1-(2,6-difluorobenzoyl)-3-(4-chlorophenyl)urea. J. Agric. Food Chem. 1975, 23, (3), 359-364.
- Aryl isocyanates undergo [2+2] cycloaddition reactions with dihydrofuran to give bicyclic ß-lactams in high yield: Nippon Kagaku Kaishi, 459 (1995):
- For general chemistry of isocyanates, see Appendix 3.