Search
Thermo Scientific Chemicals
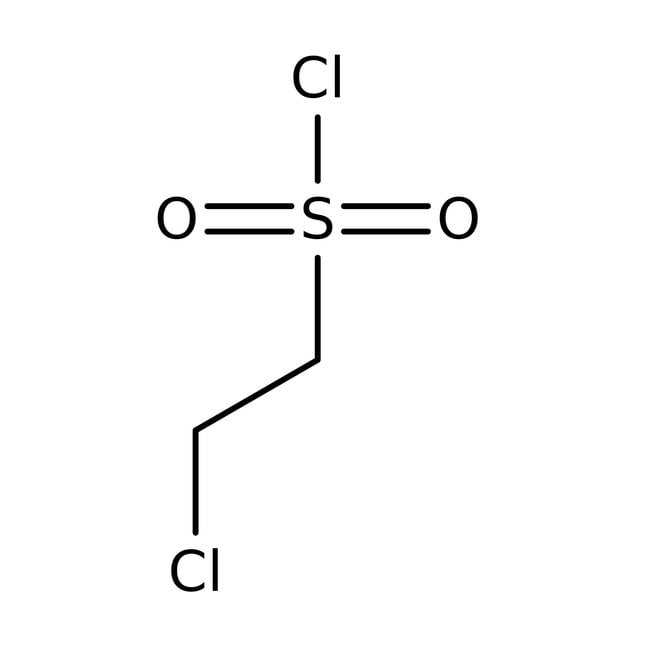
2-Chloroethanesulfonyl chloride, 98%
CAS: 1622-32-8 | C2H4Cl2O2S | 163.012 g/mol
Catalog number A15377.18
also known as A15377-18
Price (EUR)
165,00
Each
Quantity:
50 g
Price (EUR)
165,00
Each
Chemical Identifiers
CAS1622-32-8
IUPAC Name2-chloroethane-1-sulfonyl chloride
Molecular FormulaC2H4Cl2O2S
InChI KeyVHCSBTPOPKFYIU-UHFFFAOYSA-N
SMILESClCCS(Cl)(=O)=O
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Clear colorless to pale brown
FormLiquid
Assay (Titration ex Chloride)≥97.5 to ≤102.5% (non-U.S. specification)
Assay from Suppliers CofA≥97.5% (U.S. specification)
CommentSpecification differs for U.S. and non-U.S. material where indicated
View more
2-Chloroethanesulfonyl chloride was used in one-pot sulfonylation/intramolecular thia-Michael protocol for the synthesis of 1,5,2-dithiazepine 1,1-dioxides. It was also used in synthesis of vinyl sulfonamides with a furan, carbocyclic, semi cyclic or acyclic 1,3-diene moiety.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
2-Chloroethanesulfonyl chloride was used in one-pot sulfonylation/intramolecular thia-Michael protocol for the synthesis of 1,5,2-dithiazepine 1,1-dioxides. It was also used in synthesis of vinyl sulfonamides with a furan, carbocyclic, semi cyclic or acyclic 1,3-diene moiety.
Solubility
Reacts with water.
Notes
Moisture sensitive. Store under inert gas. Store away from moisture, oxidizing agents. Store in refrigerator.
2-Chloroethanesulfonyl chloride was used in one-pot sulfonylation/intramolecular thia-Michael protocol for the synthesis of 1,5,2-dithiazepine 1,1-dioxides. It was also used in synthesis of vinyl sulfonamides with a furan, carbocyclic, semi cyclic or acyclic 1,3-diene moiety.
Solubility
Reacts with water.
Notes
Moisture sensitive. Store under inert gas. Store away from moisture, oxidizing agents. Store in refrigerator.
RUO – Research Use Only
General References:
- James Frederick King; Kishan Chand Khemani. The mechanism of hydrolysis of 2-hydroxyethanesulfonyl chloride: the intermediacy of 1,2-oxathietane 2,2-dioxide (β-sultone). Canadian Journal of Chemistry. 1989, 67 (12), 2162-2172.
- C. W. Huffman. Formylation of Amines. J. Org. Chem. 1958, 23 (5), 727-729.
- In the presence of a base, converts alcohols to vinyl sulfonates: J. Am. Chem. Soc., 68, 1797 (1946); Angew. Chem. Int. Ed., 4, 300 (1965). These electrophilic alkenes readily undergo conjugate addition reactions with various nucleophiles:
- Reaction with pentafluorophenol, in the presence of Et3N, gives PFP vinylsulfonate, which can be converted to a sulfonamide via tin-mediated radical addition of alkyl halides to the olefinic bond, followed by displacement of the PFP group with an amine: Org. Lett., 42549 (2002).