Search
Thermo Scientific Chemicals
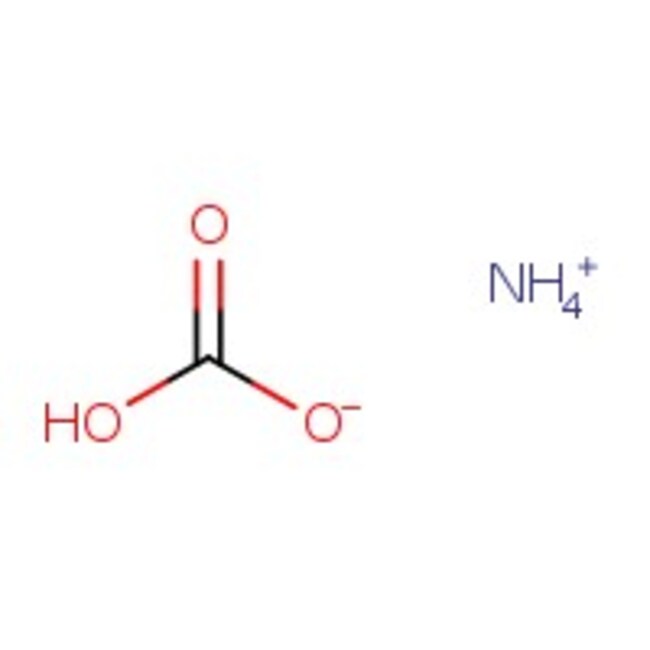
Ammonium hydrogen carbonate, 98%
CAS: 1066-33-7 | C3H8N2O9Zn2 | 346.86 g/mol
Catalog number A18566.36
also known as A18566-36
Price (EUR)
26,50
Each
Quantity:
500 g
Price (EUR)
26,50
Each
Chemical Identifiers
CAS1066-33-7
IUPAC Namecarbonic acid amine
Molecular FormulaCH5NO3
InChI KeyATRRKUHOCOJYRX-UHFFFAOYSA-N
SMILESN.OC(O)=O
View more
Specifications Specification Sheet
Specification Sheet
FormCrystals or powder or crystalline powder or fused solid
Assay (Aqueous acid-base Titration)≥97.5 to ≤102.5%
Identification (FTIR)Conforms
Appearance (Color)White
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Suitable for battery materials development.
Applications
Ammonium bicarbonate is a commonly used reagent for industrial and research procedures. It acts a good buffer in lyophilization and matrix assisted laser desorption. It is also utilized for the in-gel digestion of proteins by trypsin and in the MALDI mass spectrometric analysis of proteins. It is also used to make other ammonium compounds, in food processing, and for other uses. Ammonium bicarbonate can be used to study biological buffers. It is also used in a study that demonstrated that ammonium bicarbonate salts, which can be regenerated using low-temperature waste heat, can also produce sufficient voltage for hydrogen gas generation in a microbial reverse-electrodialysis electrolysis cells. It has also been used in a study that developed a fast and sensitive method for the simultaneous determination of Sudan dyes in food samples using partial filling micellar electrokinectic chromatography-mass spectrometry.
Notes
Store away from oxidizing agents, acids and bases. Keep the container tightly closed and place it in a cool, dry and well ventilated condition.
Ammonium bicarbonate is a commonly used reagent for industrial and research procedures. It acts a good buffer in lyophilization and matrix assisted laser desorption. It is also utilized for the in-gel digestion of proteins by trypsin and in the MALDI mass spectrometric analysis of proteins. It is also used to make other ammonium compounds, in food processing, and for other uses. Ammonium bicarbonate can be used to study biological buffers. It is also used in a study that demonstrated that ammonium bicarbonate salts, which can be regenerated using low-temperature waste heat, can also produce sufficient voltage for hydrogen gas generation in a microbial reverse-electrodialysis electrolysis cells. It has also been used in a study that developed a fast and sensitive method for the simultaneous determination of Sudan dyes in food samples using partial filling micellar electrokinectic chromatography-mass spectrometry.
Notes
Store away from oxidizing agents, acids and bases. Keep the container tightly closed and place it in a cool, dry and well ventilated condition.
RUO – Research Use Only
General References:
- Ji Guang Lia.; Takayasu Ikegami.; Jong-Heun Lee.; Toshiyuki Mori.; and Yoshiyuki Yajima. Reactive yttrium aluminate garnet powder via coprecipitation using ammonium hydrogen carbonate as the precipitant.J. Mater. Res.2000,15(9), 1864-1867.
- Kaboudin Babak.; Saadati Fariba. Studies on the synthesis of 1-aminoalkylphosphonates from aldehydes using silica-supported ammonium hydrogen carbonate.Synthesis,1969,1249-1252.