Search
Gibco™
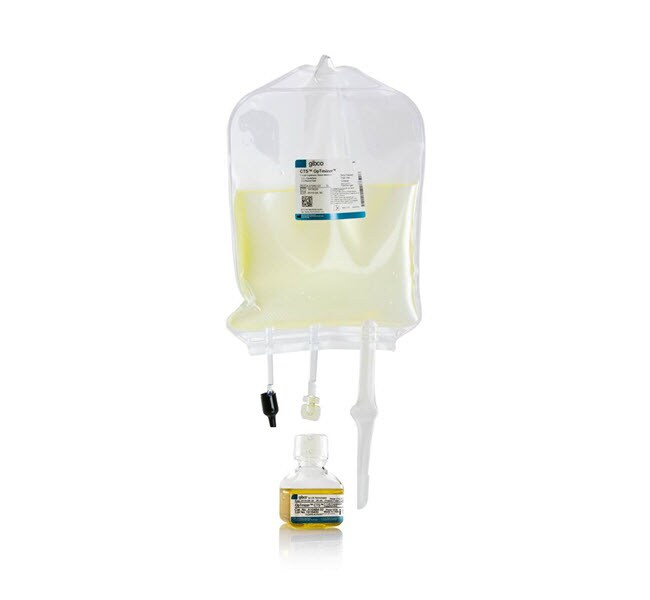
CTS™ OpTmizer™ T-Cell Expansion SFM, sin rojo fenol, formato de bolsa
SFM de expansión de células T CTS OpTmizer Gibco que favorece el crecimiento y la expansión de linfocitos T humanos.SeMás información
| Número de catálogo | Cantidad |
|---|---|
| A3705003 | 1000 mL |
Número de catálogo A3705003
Precio (USD)
-
Cantidad:
1000 mL
SFM de expansión de células T CTS OpTmizer Gibco que favorece el crecimiento y la expansión de linfocitos T humanos.Se trata de un medio completo sin suero ni xenógenos compuesto de medio basal de expansión de linfocitos T CTS OpTmizer y suplemento de expansión de linfocitos T CTS OpTmizer, que se mezclan antes de usar.
El SFM de expansión de linfocitos T CTS OpTmizer está disponible con y sin rojo de fenol.Esta versión del SFM CTS OpTmizer carece de rojo de fenol y está disponible en formato de frasco y bolsa.
Entre las características del SFM de expansión de linfocitos T CTS OpTmizer se incluyen:
•Favorece el cultivo de linfocitos T de alta densidad (>4 x 106 linfocitos T CD3+/ml)
• Favorece la activación de linfocitos T mediante gránulos magnéticos Dynabeads, anticuerpos solubles y protocolos de células presentadoras de anticuerpos estimulantes
• Fenotipo similar, función (p. ej., perfil de secreción de citoquinas) y viabilidad de los linfocitos T cultivados con medio suplementado con suero AB humano convencional
• Favorece un fenotipo de linfocitos T similar al medio suplementado con suero humano
• Demuestra una mayor eficacia y persistencia de las células CART-19 cuando se cultivan en un medio suplementado con el sustituto de suero de células inmunes CTS (ICSR)
Rendimiento funcional demostrado
Las células cultivadas en el SFM de expansión de linfocitos T CTS OpTmizer con o sin suero humano demuestran buenos perfiles de producción de citoquinas en linfocitos T activados policlonales.El SFM de expansión de linfocitos T CTS OpTmizer mantiene el fenotipo de linfocitos T de memoria central CD62L+ deseado.
Fabricado según las GMP
El sitio de fabricación principal para el SFM de expansión de linfocitos T CTS OpTmizer está registrado por la FDA y cuenta con un sistema de gestión de calidad certificado por la norma ISO13485.Los métodos y controles utilizados para la fabricación están en conformidad con las buenas prácticas de fabricación actuales para productos sanitarios, norma 21 CFR parte 820, de la normativa.Existen sitios de fabricación alternativos que proporcionan una mitigación de riesgos al ayudar a garantizar la seguridad del suministro.
Versatilidad
El SFM de expansión de linfocitos T CTS OpTmizer se ha adoptado ampliamente en aplicaciones clínicas de investigación de linfocitos T, incluido el cultivo de linfocitos T CAR.Funciona con varios formatos de cultivo, incluidos matraces de cultivo, bolsas de cultivo y sistemas de biorreactores de balanceo.El SFM de expansión de linfocitos T CTS OpTmizer está diseñado para favorecer la expansión de linfocitos T humanos cultivados sin suplemento de suero adicional.Generalmente se observa un mayor rendimiento cuando se complementa con el sustituto de suero de células inmunes CTS.
Lo que le ofrece
El SFM de expansión de linfocitos T CTS OpTmizer incluye dos componentes:1 litro de medio basal de expansión de linfocitos T CTS OpTmizer y 26 ml de medio concentrado auxiliar de expansión de linfocitos T CTS OpTmizer, que se mezclan para crear el medio completo.Disponibilidad de documentación de trazabilidad completa, como certificado de origen y certificado de análisis.
El SFM de expansión de linfocitos T CTS OpTmizer está disponible con y sin rojo de fenol.Esta versión del SFM CTS OpTmizer carece de rojo de fenol y está disponible en formato de frasco y bolsa.
Entre las características del SFM de expansión de linfocitos T CTS OpTmizer se incluyen:
•Favorece el cultivo de linfocitos T de alta densidad (>4 x 106 linfocitos T CD3+/ml)
• Favorece la activación de linfocitos T mediante gránulos magnéticos Dynabeads, anticuerpos solubles y protocolos de células presentadoras de anticuerpos estimulantes
• Fenotipo similar, función (p. ej., perfil de secreción de citoquinas) y viabilidad de los linfocitos T cultivados con medio suplementado con suero AB humano convencional
• Favorece un fenotipo de linfocitos T similar al medio suplementado con suero humano
• Demuestra una mayor eficacia y persistencia de las células CART-19 cuando se cultivan en un medio suplementado con el sustituto de suero de células inmunes CTS (ICSR)
Rendimiento funcional demostrado
Las células cultivadas en el SFM de expansión de linfocitos T CTS OpTmizer con o sin suero humano demuestran buenos perfiles de producción de citoquinas en linfocitos T activados policlonales.El SFM de expansión de linfocitos T CTS OpTmizer mantiene el fenotipo de linfocitos T de memoria central CD62L+ deseado.
Fabricado según las GMP
El sitio de fabricación principal para el SFM de expansión de linfocitos T CTS OpTmizer está registrado por la FDA y cuenta con un sistema de gestión de calidad certificado por la norma ISO13485.Los métodos y controles utilizados para la fabricación están en conformidad con las buenas prácticas de fabricación actuales para productos sanitarios, norma 21 CFR parte 820, de la normativa.Existen sitios de fabricación alternativos que proporcionan una mitigación de riesgos al ayudar a garantizar la seguridad del suministro.
Versatilidad
El SFM de expansión de linfocitos T CTS OpTmizer se ha adoptado ampliamente en aplicaciones clínicas de investigación de linfocitos T, incluido el cultivo de linfocitos T CAR.Funciona con varios formatos de cultivo, incluidos matraces de cultivo, bolsas de cultivo y sistemas de biorreactores de balanceo.El SFM de expansión de linfocitos T CTS OpTmizer está diseñado para favorecer la expansión de linfocitos T humanos cultivados sin suplemento de suero adicional.Generalmente se observa un mayor rendimiento cuando se complementa con el sustituto de suero de células inmunes CTS.
Lo que le ofrece
El SFM de expansión de linfocitos T CTS OpTmizer incluye dos componentes:1 litro de medio basal de expansión de linfocitos T CTS OpTmizer y 26 ml de medio concentrado auxiliar de expansión de linfocitos T CTS OpTmizer, que se mezclan para crear el medio completo.Disponibilidad de documentación de trazabilidad completa, como certificado de origen y certificado de análisis.
Especificaciones
Tipo de célulaCélulas T
Línea de productosOpTmizer
Tipo de productoMedio sin suero (SFM)
Cantidad1000 mL
Condiciones de envíoTemperatura ambiente
Sin aditivosSin rojo fenol
Unit SizeEach
Contenido y almacenamiento
• Bolsa de 1 x 1000 ml CTS OpTmizer Expansion Basal Medium, almacenar de 2°a 8°C, proteger de
• luz 1 x 26 ml CTS OpTmizer Expansion Supplement, almacenar en la oscuridad (de 2°a 8°C.)
• luz 1 x 26 ml CTS OpTmizer Expansion Supplement, almacenar en la oscuridad (de 2°a 8°C.)