Search
Alfa Aesar
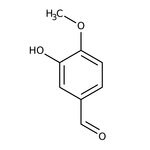
3-Hydroxy-4-methoxybenzaldehyde, 98%
CAS: 621-59-0 | C8H8O3 | 152.149 g/mol
Catalog number ALF-A12866-06
HSN 29124999
Price (INR)
2,210.00
Each
Quantity:
5 g
Price (INR)
2,210.00
Each
Chemical Identifiers
CAS621-59-0
IUPAC Name3-hydroxy-4-methoxybenzaldehyde
Molecular FormulaC8H8O3
InChI KeyJVTZFYYHCGSXJV-UHFFFAOYSA-N
SMILESCOC1=CC=C(C=O)C=C1O
View more
Specifications Specification Sheet
Specification Sheet
FormCrystals or powder or crystalline powder
Melting Point (clear melt)111.0-117.0°C
Assay (GC)≥97.5%
Appearance (Color)White to cream to gray to pale brown
3-Hydroxy-4-methoxybenzaldehyde acts as a precursor for the stereoselective synthesis of the anticancer drug (Z)-combretastatin A-4 and glycitein. It also used as an important raw material for the preparation of morphine. Further, it is involved in the preparation of Schiff- bases by reacting with furan-2-carboxylic acid hydrazide and thiophene-2-carboxylic acid hydrazide. In addition to this, it is used to prepare (Z)-2-(3-hydroxy-4-methoxybenzylidene)-1-azabicyclo[2.2.2]octan-3-one by reacting with with1-azabicyclo[2.2.2]octan-3-one.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
3-Hydroxy-4-methoxybenzaldehyde acts as a precursor for the stereoselective synthesis of the anticancer drug (Z)-combretastatin A-4 and glycitein. It also used as an important raw material for the preparation of morphine. Further, it is involved in the preparation of Schiff- bases by reacting with furan-2-carboxylic acid hydrazide and thiophene-2-carboxylic acid hydrazide. In addition to this, it is used to prepare (Z)-2-(3-hydroxy-4-methoxybenzylidene)-1-azabicyclo[2.2.2]octan-3-one by reacting with with1-azabicyclo[2.2.2]octan-3-one.
Solubility
Soluble in acetone and methanol.
Notes
Air sensitive. Store in a cool place. Incompatible with strong oxidizing agents and strong bases.
3-Hydroxy-4-methoxybenzaldehyde acts as a precursor for the stereoselective synthesis of the anticancer drug (Z)-combretastatin A-4 and glycitein. It also used as an important raw material for the preparation of morphine. Further, it is involved in the preparation of Schiff- bases by reacting with furan-2-carboxylic acid hydrazide and thiophene-2-carboxylic acid hydrazide. In addition to this, it is used to prepare (Z)-2-(3-hydroxy-4-methoxybenzylidene)-1-azabicyclo[2.2.2]octan-3-one by reacting with with1-azabicyclo[2.2.2]octan-3-one.
Solubility
Soluble in acetone and methanol.
Notes
Air sensitive. Store in a cool place. Incompatible with strong oxidizing agents and strong bases.
RUO – Research Use Only
General References:
- Aggarwal, R. L.; Di Cecca, S.; Farrar, L. W.; Shabshelowitz, A.; Jeys, T. H. Sensitive Detection and Identification of Isovanillin Aerosol Particles at the pg/cm3 Mass Concentration Level Using Raman Spectroscopy. Aerosol Sci. Tech. 2015, 49 (9), 753-756.
- Vusovich, O. V.; Lapin, I. N.; Svetlichnyi, V. A.; Sul’timova, N. B.; Tchaikovskaya, O. N. Comparison of Vanillin and Isovanillin Photolysis in Aqueous Solutions. Russ. Phys. J. 2014, 56 (11), 1287-1291.