Search
Thermo Scientific Chemicals
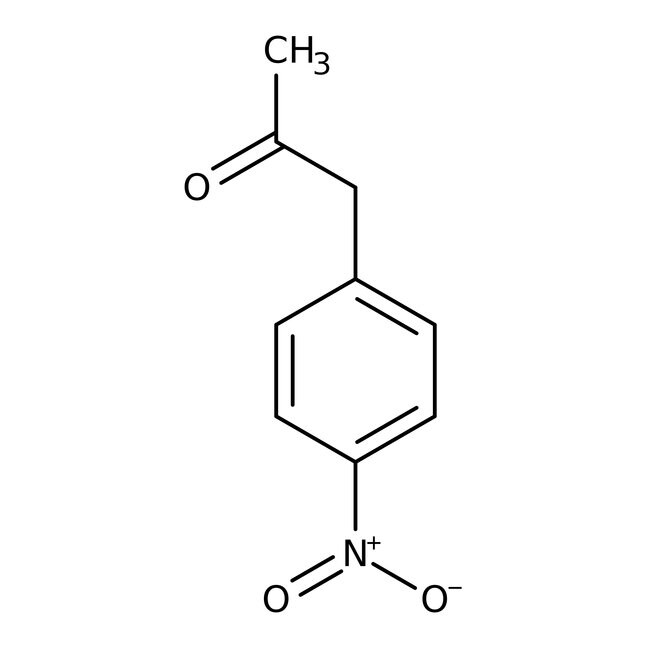
4-Nitrophenylacetone, 98%
CAS: 5332-96-7 | C9H9NO3 | 179.175 g/mol
Catalog number B21183.06
also known as B21183-06
Price (EUR)
208,00
Each
Quantity:
5 g
Price (EUR)
208,00
Each
Chemical Identifiers
CAS5332-96-7
IUPAC Name1-(4-nitrophenyl)propan-2-one
Molecular FormulaC9H9NO3
InChI KeyGEWWCWZGHNIUBW-UHFFFAOYSA-N
SMILESCC(=O)CC1=CC=C(C=C1)[N+]([O-])=O
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Yellow to orange
FormCrystals or powder or crystalline powder
Assay (GC)≥97.5%
Identification (FTIR)Conforms
Melting Point (clear melt)60.0-67.0?C
It is applied as an intermediate in organic synthesis.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
It is applied as an intermediate in organic synthesis.
Solubility
Insoluble in water, soluble in usual organic solvents.
Notes
Store away from oxidizing agents. Keep the container tightly closed and place it in a cool, dry and well ventilated condition.
It is applied as an intermediate in organic synthesis.
Solubility
Insoluble in water, soluble in usual organic solvents.
Notes
Store away from oxidizing agents. Keep the container tightly closed and place it in a cool, dry and well ventilated condition.
RUO – Research Use Only
General References:
- John Emsley, et al. β-Diketone interactions. Part 8. The hydrogen bonding of the enol tautomers of some 3-substituted pentane-2,4-diones.J. Chem. Soc., Perkin Trans. 2,1989(5), 527-533.
- Zhiqiang Guo, et al. Synthesis and structure-Activity relationships of thieno[2,3-d]pyrimidine-2,4-dione derivatives as potent GnRH receptor antagonists.Bioorg. Med. Chem. Lett.200313(20), 3617-3622.