Search
Thermo Scientific Chemicals
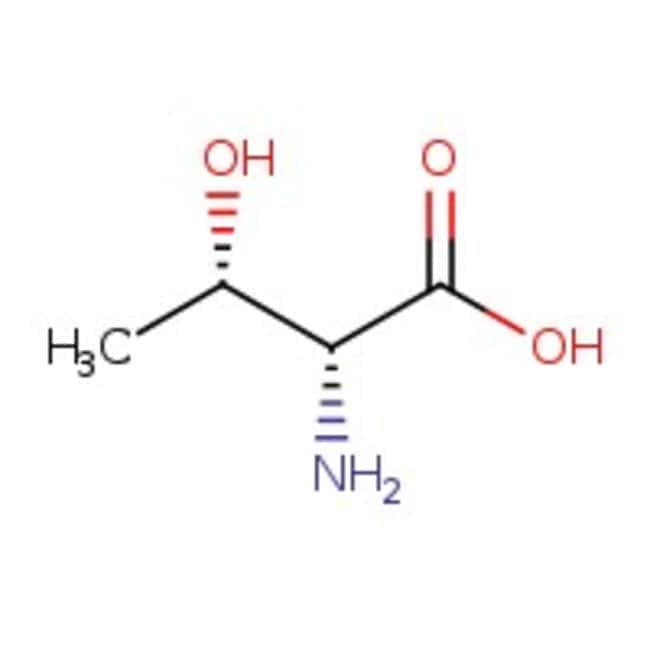
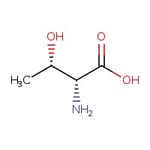
D-allo-treonina, 99 %, Thermo Scientific Chemicals
CAS: 24830-94-2 | C4H9NO3 | 119.12 g/mol
Número de catálogo H27050.03
también denominado H27050-03
Precio (USD)
-
Cantidad:
1 g
Identificadores químicos
CAS24830-94-2
IUPAC Name(2R,3S)-2-amino-3-hydroxybutanoic acid
Molecular FormulaC4H9NO3
InChI KeyAYFVYJQAPQTCCC-STHAYSLISA-N
SMILESC[C@H](O)[C@@H](N)C(O)=O
Ver más
Especificaciones Hoja de especificaciones
Hoja de especificaciones
Appearance (Color)White
FormCrystals or powder or crystalline powder
Assay (Non-aqueous acid-base Titration)≥98.5 to ≤101.5%
Optical Rotation-8.7 ± 1? (c=2 in water)
They are found as constituents of an increasing group of biologically active peptides. Stereocontrolled syntheses of chiral and racemic key intermediates to thienamycin from d-allo-threonine and trans-crotonic acid.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Aplicaciones
Se encuentra como componentes de un grupo creciente de péptidos biológicamente activos. Stereocontrolled syntheses of chiral and racemic key intermediates to thienamycin from d-allo-threonine and trans-crotonic acid.
Solubilidad
Soluble en agua.
Notas
Almacenar en un lugar fresco. Mantenga el recipiente bien cerrado en un lugar seco y bien ventilado. Almacenar alejado de agentes oxidantes fuertes.
Se encuentra como componentes de un grupo creciente de péptidos biológicamente activos. Stereocontrolled syntheses of chiral and racemic key intermediates to thienamycin from d-allo-threonine and trans-crotonic acid.
Solubilidad
Soluble en agua.
Notas
Almacenar en un lugar fresco. Mantenga el recipiente bien cerrado en un lugar seco y bien ventilado. Almacenar alejado de agentes oxidantes fuertes.
RUO – Research Use Only
General References:
- Peter Wipf,; Chris P. Miller. Stereospecific synthesis of peptide analogs with allo-threonine and D-allo-threonine residues . J. Org. Chem.. 1993, 58 (6),1575-1578.
- Masao Shiozaki,; Noboru Ishida,; Hiroshi Maruyama,; Tetsuo Hiraoka. Stereocontrolled syntheses of chiral and racemic key intermediates to thienamycin from d-allo-threonine and trans-crotonic acid. Tetrahedron. 1983, 39(14),2399-2407.