Search
Thermo Scientific Chemicals
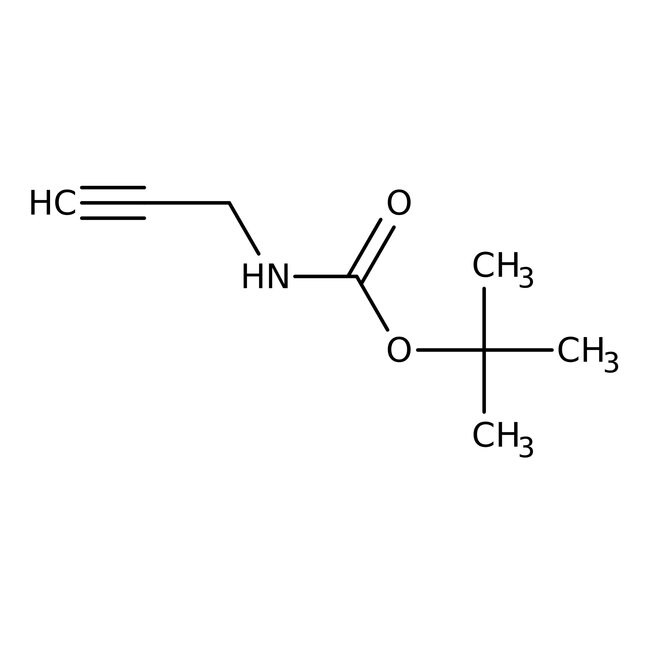
N-Boc-propargylamine, 97%
CAS: 92136-39-5 | C8H13NO2 | 155.197 g/mol
Catalog number H53406.06
also known as H53406-06
Price (EUR)
118,00
Each
Quantity:
5 g
Price (EUR)
118,00
Each
Chemical Identifiers
CAS92136-39-5
IUPAC Nametert-butyl N-(prop-2-yn-1-yl)carbamate
Molecular FormulaC8H13NO2
InChI KeyDSPYCWLYGXGJNJ-UHFFFAOYSA-N
SMILESCC(C)(C)OC(=O)NCC#C
View more
Specifications Specification Sheet
Specification Sheet
FormSolid
Assay (GC)>97%
Identification (FTIR)Conforms to structure
Appearance (Color)Yellow to brown
N-Boc-propargylamine is used to prepare triazolobenzylidene-thiazolopyrimidines. which act as CDC25 phosphatase inhibitors. Further, it is used for the synthesis of beta-glucan polysaccharide analogs. In addition to this, it is involved in the Pauson-Khand (PK) reaction of norbornadiene and N-Boc-propargylamine to prepare 4,5-disubstituted cyclopentenones .
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
N-Boc-propargylamine is used to prepare triazolobenzylidene-thiazolopyrimidines. which act as CDC25 phosphatase inhibitors. Further, it is used for the synthesis of beta-glucan polysaccharide analogs. In addition to this, it is involved in the Pauson-Khand (PK) reaction of norbornadiene and N-Boc-propargylamine to prepare 4,5-disubstituted cyclopentenones .
Solubility
Soluble in chloroform.
Notes
Moisture sensitive. Incompatible with oxidizing agents. Store in a cool place.
N-Boc-propargylamine is used to prepare triazolobenzylidene-thiazolopyrimidines. which act as CDC25 phosphatase inhibitors. Further, it is used for the synthesis of beta-glucan polysaccharide analogs. In addition to this, it is involved in the Pauson-Khand (PK) reaction of norbornadiene and N-Boc-propargylamine to prepare 4,5-disubstituted cyclopentenones .
Solubility
Soluble in chloroform.
Notes
Moisture sensitive. Incompatible with oxidizing agents. Store in a cool place.
RUO – Research Use Only
General References:
- Kann, N.; Johansson, J. R.; Beke-Somfai, T. Conformational properties of 1, 4-and 1, 5-substituted 1, 2, 3-triazole amino acids-building units for peptidic foldamers. Org. Biomol. Chem. 2015, 13 (9), 2776-2785.
- Yalagala, R. S.; Yan, H. Reactions of alkynes with trisubstituted (Z)-bromoalkenyl-pinacolboronates towards the synthesis of 1, 2, 4, 6-tetrasubstituted benzenes. Tetrahedron Lett. 2014, 55 (34), 4830-4832.