Search
Thermo Scientific Chemicals
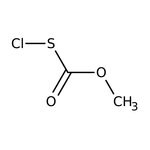
Methoxycarbonylsulfenyl chloride, 95%
CAS: 26555-40-8 | C2H3ClO2S | 126.554 g/mol
Catalog number L04350.06
also known as L04350-06
Price (EUR)
71,80
Each
Quantity:
5 g
Price (EUR)
71,80
Each
Chemical Identifiers
CAS26555-40-8
IUPAC Name(chlorosulfanyl)(methoxy)methanone
Molecular FormulaC2H3ClO2S
InChI KeyTXJXPZVVSLAQOQ-UHFFFAOYSA-N
SMILESCOC(=O)SCl
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Clear colorless to yellow
Assay (GC)>94.0%
Refractive Index1.4795-1.4865 @ 20?C
FormLiquid
Methoxycarbonylsulfenyl chloride is used to prepare 3-[(methoxycarbonyl)dithio]-L-alanine by reacting with L-cysteine. Further, it is used in the synthesis of 3-2 pyridinyldithio propanoic acid hydrazide (PDPH). It serves as an important heterobifunctional crosslinker, which is a crosslinker used to make macromolecule-drug conjugates.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Methoxycarbonylsulfenyl chloride is used to prepare 3-[(methoxycarbonyl)dithio]-L-alanine by reacting with L-cysteine. Further, it is used in the synthesis of 3-2 pyridinyldithio propanoic acid hydrazide (PDPH). It serves as an important heterobifunctional crosslinker, which is a crosslinker used to make macromolecule-drug conjugates.
Solubility
Miscible with dichlormethane.
Notes
Moisture sensitive. Store in a cool place. Keep the container tightly closed in a dry and well-ventilated place. Incompatible with strong oxidizing agents.
Methoxycarbonylsulfenyl chloride is used to prepare 3-[(methoxycarbonyl)dithio]-L-alanine by reacting with L-cysteine. Further, it is used in the synthesis of 3-2 pyridinyldithio propanoic acid hydrazide (PDPH). It serves as an important heterobifunctional crosslinker, which is a crosslinker used to make macromolecule-drug conjugates.
Solubility
Miscible with dichlormethane.
Notes
Moisture sensitive. Store in a cool place. Keep the container tightly closed in a dry and well-ventilated place. Incompatible with strong oxidizing agents.
RUO – Research Use Only
General References:
- Reacts with arenes under Friedel-Crafts conditions to give S-aryl thiocarbonates, thus providing a method for the introduction of the thiol group into an aromatic ring: Z. Chem., 17, 411 (1977).
- Reagent for the preparation of unsymmetrical disulfides, by successive reaction with different thiols: Helv. Chim. Acta, 56, 1370 (1973):
- Reaction of the same intermediate with KO-t-Bu provides a route to trisulfides. Cyclic trisulfides can also be prepared: J. Org. Chem., 44, 4144 (1979).
- Kaushik, R.; Kumar, P.; Ghosh, A.; Gupta, N.; Kaur, D.; Arora, S.; Jose, D. A. Alizarin red S-zinc(II) fluorescent ensemble for selective detection of hydrogen sulphide and assay with an H2S donor. RSC Adv. 2015, 5 (97), 79309-79316.
- Artaud, I.; Galardon, E. A Persulfide Analogue of the Nitrosothiol SNAP: Formation, Characterization and Reactivity. ChemBioChem 2014, 15 (16), 2361-2364.