Search
Thermo Scientific Chemicals
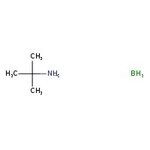
Borane-tert-butylamine complex, 97%
CAS: 7337-45-3 | C4H14BN | 86.97 g/mol
Catalog number L13177.18
also known as L13177-18
Price (EUR)
116,00
Each
Quantity:
50 g
Price (EUR)
116,00
Each
Chemical Identifiers
CAS7337-45-3
IUPAC Name2-methylpropan-2-amine borane
Molecular FormulaC4H14BN
InChI KeyGKFJEDWZQZKYHV-UHFFFAOYSA-N
SMILESB.CC(C)(C)N
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)White
Assay from Supplier's CofA≥96.0 to ≤104.0%
FormCrystalline powder or crystals
Identification (FTIR)Conforms (non-U.S. specification)
Borane-tert-butylamine complex is used in stereoselective reduction of a steroidal ketone and for a review of amine boranes as selective reducing and hydroborating agents. It is also used as strong co-reducing agent in the syntheses of copper (I) oxide nanoparticles, diphosphine-protected gold nanocluster and 4-acetoxycinnamyl alcohols.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Borane-tert-butylamine complex is used in stereoselective reduction of a steroidal ketone and for a review of amine boranes as selective reducing and hydroborating agents. It is also used as strong co-reducing agent in the syntheses of copper (I) oxide nanoparticles, diphosphine-protected gold nanocluster and 4-acetoxycinnamyl alcohols.
Solubility
Soluble in water (27 g/L at 20°C) and methanol.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Incompatible with oxidizing agents and acids.
Borane-tert-butylamine complex is used in stereoselective reduction of a steroidal ketone and for a review of amine boranes as selective reducing and hydroborating agents. It is also used as strong co-reducing agent in the syntheses of copper (I) oxide nanoparticles, diphosphine-protected gold nanocluster and 4-acetoxycinnamyl alcohols.
Solubility
Soluble in water (27 g/L at 20°C) and methanol.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Incompatible with oxidizing agents and acids.
RUO – Research Use Only
General References:
- John M Pettibone.; Jeffrey W Hudgens. Reaction network governing diphosphine-protected gold nanocluster formation from nascent cationic platforms. Physical Chemistry Chemical Physics. 2012, 14 (12), 4142-4154.
- Na Rae Kim.; Inyu Jung.; Yun Hwan Jo.; Hyuck Mo Lee. A simple process for the preparation of copper (I) oxide nanoparticles by a thermal decomposition process with borane tert-butylamine complex. Journal of Nanoscience and Nanotechnology. 2013, 13 (9), 6027-6032.
- Solid, water-soluble reducing agent. For use in stereoselective reduction of a steroidal ketone, see: Synth. Commun., 11, 875 (1981). Reduces aldehydes, generated in situ by hydrolysis of acetals with aqueous triflic acid in THF, to primary alcohols: Synlett, 59 (1999). For a review of amine boranes as selective reducing and hydroborating agents, see: Org. Prep. Proced. Int., 16, 335 (1984).