Search
Thermo Scientific Chemicals
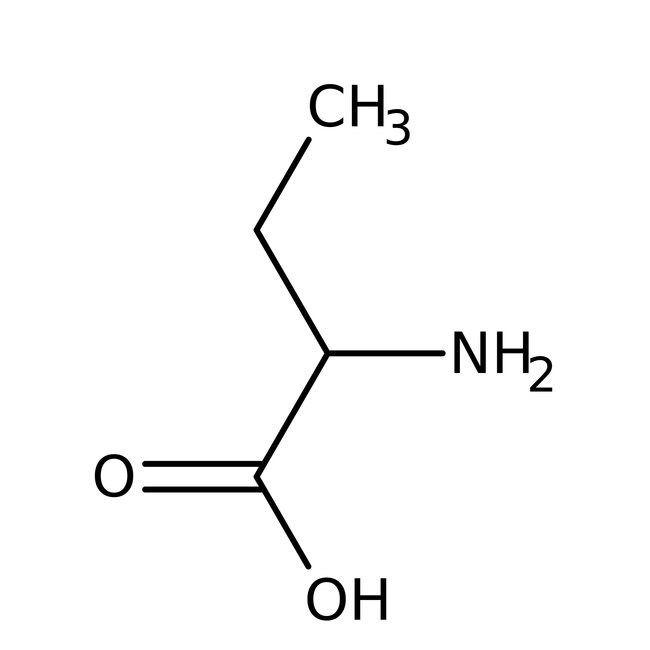
L-(+)-2-Aminobutyric acid, 98+%
CAS: 1492-24-6 | C4H9NO2 | 103.121 g/mol
Catalog number L13984.03
also known as L13984-03
Price (TWD)Request A Quote
-
Quantity:
1 g
Chemical Identifiers
CAS1492-24-6
IUPAC Name2-aminobutanoic acid
Molecular FormulaC4H9NO2
InChI KeyQWCKQJZIFLGMSD-UHFFFAOYNA-N
SMILESCCC(N)C(O)=O
View more
Specifications Specification Sheet
Specification Sheet
Assay (Non-aqueous acid-base Titration)≥98.0 to ≤102.0%
FormCrystals or powder or crystalline powder or flakes
Optical Rotation20.8 ± 1.0° (c=4.8 in 5N HCl)
Appearance (Color)White to pale cream to pale brown
L-(+)-2-Aminobutyric acid is used in the biosynthesis of nonribosomal peptides. It acts as a receptor antagonist. It is also used as a chiral reagent. Further, it is used in the determination of substrate of glutamyl cysteine acid synthase. In addition to this, it is also utilized as a drug intermediate.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
L-(+)-2-Aminobutyric acid is used in the biosynthesis of nonribosomal peptides. It acts as a receptor antagonist. It is also used as a chiral reagent. Further, it is used in the determination of substrate of glutamyl cysteine acid synthase. In addition to this, it is also utilized as a drug intermediate.
Solubility
Soluble in water and acetic acid. Slightly soluble in alcohol and ether.
Notes
Incompatible with strong oxidizing agents.
L-(+)-2-Aminobutyric acid is used in the biosynthesis of nonribosomal peptides. It acts as a receptor antagonist. It is also used as a chiral reagent. Further, it is used in the determination of substrate of glutamyl cysteine acid synthase. In addition to this, it is also utilized as a drug intermediate.
Solubility
Soluble in water and acetic acid. Slightly soluble in alcohol and ether.
Notes
Incompatible with strong oxidizing agents.
RUO – Research Use Only
General References:
- Martinez, S.; Hausinger, R. P. Catalytic Mechanisms of Fe(II)- and 2-Oxoglutarate-dependent Oxygenases. J. Biol. Chem. 2015, 290 (34), 20702-20711.
- Soriano-Maldonado, P.; Las Heras-Vazquez, F. J.; Clemente-Jimenez, J. M.; Rodriguez-Vico, F.; Martínez-Rodríguez, S. Enzymatic dynamic kinetic resolution of racemic N-formyl- and N-carbamoyl-amino acids using immobilized l-N-carbamoylase and N-succinyl-amino acid racemase. Appl. Microbiol. Biotechnol. 2015, 99 (1), 283-291.