Search
Thermo Scientific Chemicals
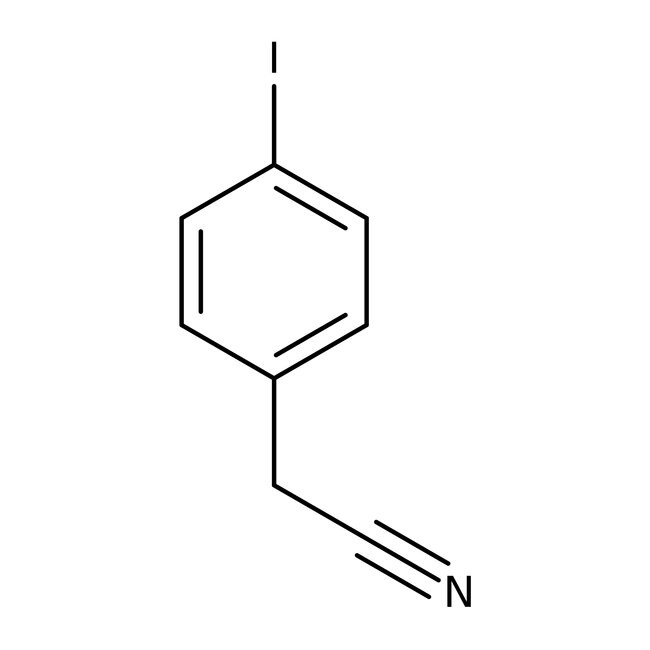
4-Iodophenylacetonitrile, 97%
CAS: 51628-12-7 | C8H6IN | 243.047 g/mol
| Catalog Number | Quantity |
|---|---|
| L15119.03 also known as L15119-03 | 1 g |
Catalog number L15119.03
also known as L15119-03
Price (USD)
-
Quantity:
1 g
Chemical Identifiers
CAS51628-12-7
IUPAC Name2-(4-iodophenyl)acetonitrile
Molecular FormulaC8H6IN
InChI KeyPNXWQTYSBFGIFD-UHFFFAOYSA-N
SMILESIC1=CC=C(CC#N)C=C1
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)White to cream or yellow
Assay (GC)> 96.0%
FormCrystalline or fused solid or powder
4-Iodophenylacetonitrile is used as an intermediate in organic syntheses and in pharmaceuticals.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
4-Iodophenylacetonitrile is used as an intermediate in organic syntheses and in pharmaceuticals.
Solubility
Insoluble in water
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. It is sensitive to moisture. Incompatible with oxidizing agents.
4-Iodophenylacetonitrile is used as an intermediate in organic syntheses and in pharmaceuticals.
Solubility
Insoluble in water
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. It is sensitive to moisture. Incompatible with oxidizing agents.
RUO – Research Use Only
General References:
- Steven K. Pollack.; Jawad Naciri.; John Mastrangelo.; Charles H. Patterson.; Johnny Torres.; Martin Moore.; Ranganathan Shashidhar.; James G. Kushmerick. Sequential Deprotection for Control of Orientation in the Self-Assembly of Asymmetric Molecules for Molecular Electronic Devices. Langmuir. 2004, 20 (5), 1838-1842.
- Jeremy J. Clemens.; Juliana L. Asgian.; Brett B. Busch.; Timothy Coon.; Justin Ernst.; Leonard Kaljevic.; Paul J. Krenitsky.; Timothy D. Neubert.; Edwin J. Schweiger.; Andreas Termin.; Dean Stamos. Diastereoselective One-Pot Knoevenagel Condensation/Corey-Chaykovsky Cyclopropanation. J. Org. Chem. 2013, 78 (2), 780-785.