Search
Thermo Scientific Chemicals
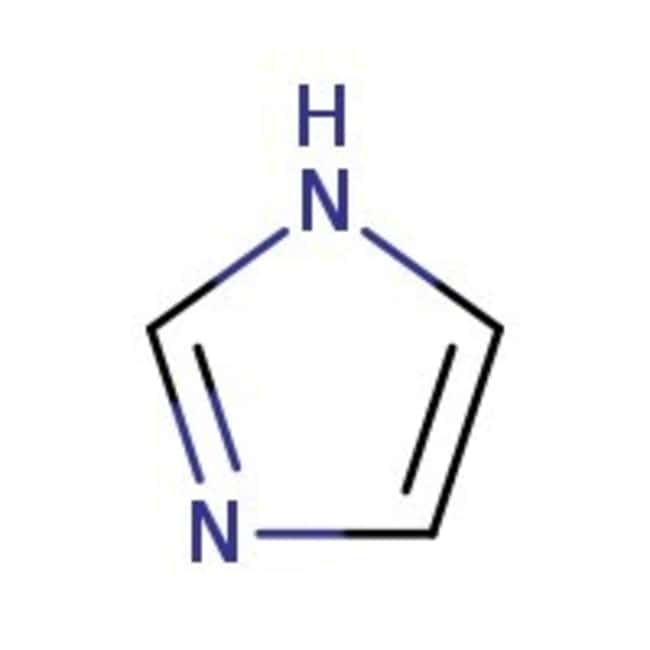
Imidazol, 99 %, Thermo Scientific Chemicals
CAS: 288-32-4 | C3H4N2 | 68.08 g/mol
Número de catálogo A10221.22
también denominado A10221-22
Precio (USD)
-
Cantidad:
100 g
Identificadores químicos
CAS288-32-4
IUPAC Name1H-imidazole
Molecular FormulaC3H4N2
InChI KeyRAXXELZNTBOGNW-UHFFFAOYSA-N
SMILESN1C=CN=C1
Ver más
Especificaciones Hoja de especificaciones
Hoja de especificaciones
CommentPurchased in the U.K. and in other countries
Water Content (Karl Fischer Titration)≤0.5%
Assay from Suppliers CofA≥98.5 to ≤101.5% (Acidimetric) (non-U.K. specification)
Appearance (Color)White to pale cream to pale yellow
FormCrystals or flakes or powder or crystalline flakes or crystalline powder
Ver más
Imidazole is used as a buffer in the range of pH 6.2-7.8. It is also an histamine antagonist. It acts as a chelator and forms complexes with various divalent cations. It is used as a corrosion inhibitor on certain transition metals such as copper. Its derivatives, like polybenzimidazole (PBI), act as fire retardants. It finds application in photography and electronics. Imidazole salts are used as ionic liquids and precursors to stable carbenes. Imidazole derivatives like ketoconazole, miconazole and clotrimazole are involved in the treatment of various systemic fungal infections. It is a part of the theophylline molecule, present in tea leaves and coffee beans, which stimulates the central nervous system.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Aplicaciones
El imidazol se utiliza como tampón en el rango de pH 6.2-7.8. También es un antagonista de la histamina. Actúa como un quelante y forma complejos con varios cationes divalentes. Se utiliza como inhibidor de corrosión en ciertos metales de transición como el cobre. Sus derivados, como el polibencimidazol (PBI), actúan como retardantes del fuego. Encuentra aplicación en fotografía y electrónica. Las sales de imidazol se utilizan como líquidos iónicos y precursores de carbenos estables. Los derivados del imidazol como ketoconazol, miconazol y clotrimazol están involucrados en el tratamiento de varias infecciones fúngicas sistémicas. Es una parte de la molécula de teofilina, presente en hojas de té y granos de café, que estimula el sistema nervioso central.
Solubilidad
Miscible con agua, etanol, éter, acetona, cloroformo, piridina y metanol. Ligeramente miscible con benceno y éter de petróleo.
Notas
Incompatible con ácidos, ácidos anhídridos y agentes oxidantes fuertes.
El imidazol se utiliza como tampón en el rango de pH 6.2-7.8. También es un antagonista de la histamina. Actúa como un quelante y forma complejos con varios cationes divalentes. Se utiliza como inhibidor de corrosión en ciertos metales de transición como el cobre. Sus derivados, como el polibencimidazol (PBI), actúan como retardantes del fuego. Encuentra aplicación en fotografía y electrónica. Las sales de imidazol se utilizan como líquidos iónicos y precursores de carbenos estables. Los derivados del imidazol como ketoconazol, miconazol y clotrimazol están involucrados en el tratamiento de varias infecciones fúngicas sistémicas. Es una parte de la molécula de teofilina, presente en hojas de té y granos de café, que estimula el sistema nervioso central.
Solubilidad
Miscible con agua, etanol, éter, acetona, cloroformo, piridina y metanol. Ligeramente miscible con benceno y éter de petróleo.
Notas
Incompatible con ácidos, ácidos anhídridos y agentes oxidantes fuertes.
RUO – Research Use Only
General References:
- Nucleophilic catalyst for many acylation and silylation reactions, compare 4-(Dimethyl amino) pyridine, A13016.
- For use in: silylation of 1,3-diketones, see Hexamethyl disilazane, A15139; introduction of the TBDMS and TBDPS groups; see: tert-Butyl dimethyl chlorosilane, A13064, and tert-Butyl diphenyl chlorosilane, A12721, respectively.
- In combination with triphenylphosphine and iodine, vic-diols are converted to alkenes: Synthesis, 469 (1979), and alcohols to alkyl iodides: Synth. Commun., 20, 1473 (1990).
- With 2 moles of an aroyl halide gives, after hydrolysis, good yields of 2-aroylimidazoles: Synthesis, 675 (1978).
- Sankar, M.; Ajithkumar, T. G.; Sankar, G.; Manikandan, P. Supported imidazole as heterogeneous catalyst for the synthesis of cyclic carbonates from epoxides and CO 2. Catal. Commun. 2015, 59, 201-205.
- Hart, K. R.; Sottos, N. R.; White, S. R. Repeatable self-healing of an epoxy matrix using imidazole initiated polymerization. Polymer 2015, 67, 174-184.