Search
Thermo Scientific Chemicals
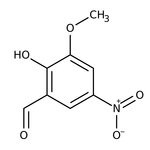
2-Hidroxi-3-metoxi-5-nitrobenzaldehído, 98 %, Thermo Scientific Chemicals
CAS: 17028-61-4 | C8H7NO5 | 197.15 g/mol
Número de catálogo A14360.03
también denominado A14360-03
Precio (USD)
-
Cantidad:
1 g
Identificadores químicos
CAS17028-61-4
Especificaciones Hoja de especificaciones
Hoja de especificaciones
Appearance (Color)Cream to yellow or brown or grey to grey/green
Melting Point (clear melt)137.0-143.0°C
Identification (FTIR)Conforms
FormPowder
Assay (GC)≥97.5%
2-Hydroxy-3-methoxy-5-nitrobenzaldehyde is used for the syntheses of zinc-selective spiropyran-based fluorescent and photoregenerable receptor, (E)-2,4-dihydroxy-N?-(2-hydroxy-3-methoxy-5-nitrobenzylidene)benzohydrazide dihydrate, ethyl 4-(8-methoxy-3?,3?-dimethyl-6-nitrospiro[chromene-2,2?-indoline]-1?-yl)butanoate and 1-(3-carbomethoxypropyl)-3-3-dimethyl-8-methoxy-6-nitrospiro[2H-1]-benzopyran-2,2-indoline.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Aplicaciones
El 2-hidroxi-3-metoxi-5-nitrobenzaldehído se utiliza para la síntesis del receptor fotoregenerable y fluorescente basado en espiropirano selectivo de cinc, (E)-2,4-dihidroxi-N′-(2-hidroxi-3-metoxi-5-nitrobencilideno)benzohidrazida dihidrato, 4-(8-metoxi-3′,3′-dimetilo-6-nitroespiro[cromeno-2,2′-indolin]-1′-il)butanoato de etilo y 1-(3-carbometoxipropilo)-3-3-dimetilo-8-metoxi-6-nitroespiro[2H-1]-benzopiran-2,2-indolina.
Solubilidad
Ligeramente soluble en agua.
Notas
Almacenar en un lugar fresco. Mantenga el recipiente bien cerrado en un lugar seco y bien ventilado. Incompatible con agentes oxidantes fuertes. Es sensible al aire.
El 2-hidroxi-3-metoxi-5-nitrobenzaldehído se utiliza para la síntesis del receptor fotoregenerable y fluorescente basado en espiropirano selectivo de cinc, (E)-2,4-dihidroxi-N′-(2-hidroxi-3-metoxi-5-nitrobencilideno)benzohidrazida dihidrato, 4-(8-metoxi-3′,3′-dimetilo-6-nitroespiro[cromeno-2,2′-indolin]-1′-il)butanoato de etilo y 1-(3-carbometoxipropilo)-3-3-dimetilo-8-metoxi-6-nitroespiro[2H-1]-benzopiran-2,2-indolina.
Solubilidad
Ligeramente soluble en agua.
Notas
Almacenar en un lugar fresco. Mantenga el recipiente bien cerrado en un lugar seco y bien ventilado. Incompatible con agentes oxidantes fuertes. Es sensible al aire.
RUO – Research Use Only
General References:
- V. Krishnakumara.; V. Balachandran. Structures and vibrational frequencies of 2-hydroxy-3-methoxy-5-nitrobenzaldehyde and 2-methoxy-1-naphthaldehyde based on density functional theory calculations. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy. 2006, 63 (2), 464-476.
- Corinne Moustrou.; André Samat.; Robert Guglielmetti.; Roger Dubest.; Francis Garnier. Synthesis of Thiophene-Substituted Spiropyrans and Spirooxazines, Precursors of Photochromic Polymers. Helvetica Chimica Acta. 1995, 78 (7), 1887-1893.