Search
Thermo Scientific Chemicals
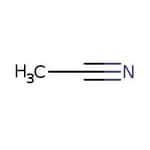
Acetonitrilo, 99 %, Thermo Scientific Chemicals
CAS: 75-05-8 | C2H3N | 41.053 g/mol
| Número de catálogo | Cantidad |
|---|---|
| A19862.AP también denominado A19862-AP | 500 mL |
Número de catálogo A19862.AP
también denominado A19862-AP
Precio (USD)
-
Cantidad:
500 mL
Identificadores químicos
CAS75-05-8
IUPAC Nameacetonitrile
Molecular FormulaC2H3N
InChI KeyWEVYAHXRMPXWCK-UHFFFAOYSA-N
SMILESCC#N
Ver más
Especificaciones Hoja de especificaciones
Hoja de especificaciones
Appearance (Color)Clear colorless
Refractive Index1.3425-1.3455 @ 20?C
Assay (GC)≥98.5%
FormLiquid
Identification (FTIR)Conforms
Acetonitrile is utilized as a polar aprotic solvent in organic synthesis, and in the purification of butadiene. It is used in all the analytical laboratories as a major component of mobile phase in High Performance Liquid Chromatography (HPLC) and in Liquid Chromatography - Mass Spectrometry (LC-MS). It is used for the extraction of fatty acids, spinning fibers and casting and molding of plastic materials. Aqueous two-phase systems based on acetonitrile and carbohydrates play an important character in the extraction and purification of biomolecules called vanillins. It acts as a stabilizer for the chlorinated solvents, and finds use in the production of DNA oligonucleotides.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Aplicaciones
El acetonitrilo se utiliza como disolvente polar aprótico en la síntesis orgánica y en la purificación del butadieno. Se utiliza en todos los laboratorios analíticos como componente principal de la fase móvil en la cromatografía de líquidos de alto rendimiento (HPLC) y en la cromatografía líquida - espectrometría de masas (CL-EM). Se utiliza para la extracción de ácidos grasos, fibras para hilatura o fundición y moldeo de materiales plásticos. Los sistemas acuosos bifásicos basados en acetonitrilo y carbohidratos desempeñan un papel importante en la extracción y purificación de las biomoléculas llamadas vanillinas. Actúa como un estabilizador para los disolventes clorados y se usa en la producción de oligonucleótidos de ADN.
Solubilidad
Miscible con agua, alcoholes, éteres, acetona, cloroformo, tetracloruro de carbono y cloruro de etileno. No miscible con hidrocarburos saturados (fracciones de petróleo).
Notas
Incompatible con metales alcalinos, ácidos, bases, agentes reductores y agentes oxidantes. Altamente inflamable. Almacenar lejos de fuentes de calor, chispas y llamas.
El acetonitrilo se utiliza como disolvente polar aprótico en la síntesis orgánica y en la purificación del butadieno. Se utiliza en todos los laboratorios analíticos como componente principal de la fase móvil en la cromatografía de líquidos de alto rendimiento (HPLC) y en la cromatografía líquida - espectrometría de masas (CL-EM). Se utiliza para la extracción de ácidos grasos, fibras para hilatura o fundición y moldeo de materiales plásticos. Los sistemas acuosos bifásicos basados en acetonitrilo y carbohidratos desempeñan un papel importante en la extracción y purificación de las biomoléculas llamadas vanillinas. Actúa como un estabilizador para los disolventes clorados y se usa en la producción de oligonucleótidos de ADN.
Solubilidad
Miscible con agua, alcoholes, éteres, acetona, cloroformo, tetracloruro de carbono y cloruro de etileno. No miscible con hidrocarburos saturados (fracciones de petróleo).
Notas
Incompatible con metales alcalinos, ácidos, bases, agentes reductores y agentes oxidantes. Altamente inflamable. Almacenar lejos de fuentes de calor, chispas y llamas.
RUO – Research Use Only
General References:
- Polar aprotic solvent for a wide variety of reactions, including nucleophilic substitutions, oxidations, reductions and organometallic reactions. Widely used with crown ethers for the generation of 'naked' anions from their salts; see, e.g. 18-Crown-6, A11249 . Preferred solvent for RuO4 oxidations, due to its coordinating ability; see Ruthenium(III) chloride hydrate, 11043 .
- Undergoes the Ritter reaction with alcohols or olefins in the presence of acid to give N-substituted acetamides. For benzylic alcohols, modified conditions employing boron trifluoride etherate give high yields: Synth. Commun., 24, 601 (1994). For an example, see 4-Methyl benzyl alcohol, A15315 .
- For use in the mild conversion of amides to nitriles, see Benzamide, A10501 .
- Cardoso, G. B.; Mourao, T.; Pereira, F. M.; Freire, M. G.; Fricks, A. T.; Soares, C. M. F.; Lima, A. S. Aqueous two-phase systems based on acetonitrile and carbohydrates and their application to the extraction of vanillin. Sep. Purif. Technol. 2013, 104 (5), 106-113.
- Xu, K.; Zhu, L.; Zhang, A.; Jiang, G.; Tang, H. A peculiar cyclic voltammetric behavior of polyaniline in acetonitrile and its application in ammonia vapor sensor. J. Electroanal. Chem. 2007, 608 (2), 141-147.
- Lam, T. W.; Zhang, H.; Siu, S. K. Reductions of Oxygen, Carbon Dioxide, and Acetonitrile by the Magnesium(II)/Magnesium(I) Couple in Aqueous Media: Theoretical Insights from a Nano-Sized Water Droplet. J. Phys. Chem. A 2015, 119 (12), 2780-2792.