Search
Thermo Scientific Chemicals
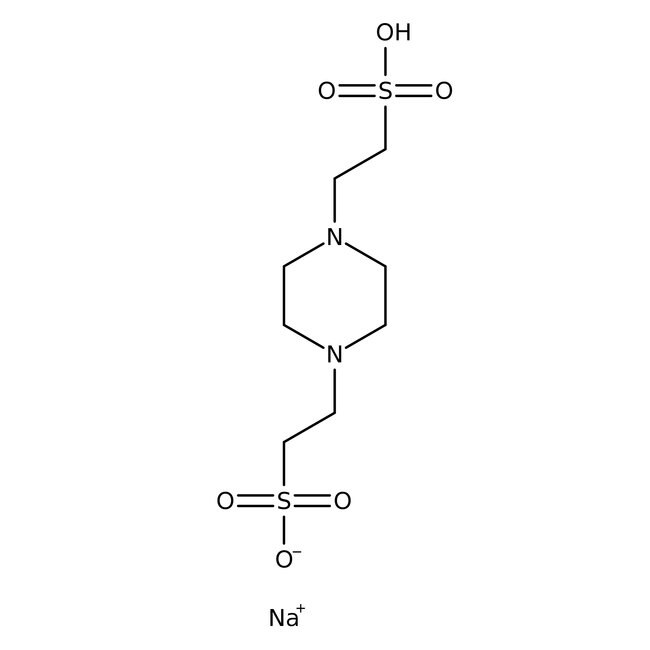
PIPES, solución de tampón de 0,5 M, pH 7,5
CAS: 5625-37-6 | C8H18N2O6S2 | 302.36 g/mol
| Número de catálogo | Cantidad |
|---|---|
| J63617.AE también denominado J63617-AE | 100 mL |
Número de catálogo J63617.AE
también denominado J63617-AE
Precio (USD)
-
Cantidad:
100 mL
Especificaciones
CAS5625-37-6
ChEBICHEBI:44933
Nombre del producto químico o materialPIPES buffer soln.
ColorIncolora
Nombre IUPAC2-[4-(2-sulfoetil)piperazina-1-il]ácido etanesulfónico
Ver más
PIPES is frequently used as a buffering agent in biochemistry. PIPES has pKa near the physiological pH which makes it useful in cell culture work. PIPES has been documented minimizing lipid loss when buffering glutaraldehyde histology in plant and animal tissues and has a negligible capacity to bind divalent ions. Fixation of fungal zoospores for fluorescence microscopy and electron microscopy was optimal with a combination of glutaraldehyde and formaldehyde in PIPES buffer is done.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
PIPES is frequently used as a buffering agent in biochemistry. PIPES has pKa near the physiological pH which makes it useful in cell culture work. PIPES has been documented minimizing lipid loss when buffering glutaraldehyde histology in plant and animal tissues and has a negligible capacity to bind divalent ions. Fixation of fungal zoospores for fluorescence microscopy and electron microscopy was optimal with a combination of glutaraldehyde and formaldehyde in PIPES buffer is done.
Solubility
Soluble in 1M NaOH (0.5 M at 20°C), water (∼1 g/L at 100°C), and 0.5 N NaOH (0.1 M).
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Keep away from strong oxidizing agents.
PIPES is frequently used as a buffering agent in biochemistry. PIPES has pKa near the physiological pH which makes it useful in cell culture work. PIPES has been documented minimizing lipid loss when buffering glutaraldehyde histology in plant and animal tissues and has a negligible capacity to bind divalent ions. Fixation of fungal zoospores for fluorescence microscopy and electron microscopy was optimal with a combination of glutaraldehyde and formaldehyde in PIPES buffer is done.
Solubility
Soluble in 1M NaOH (0.5 M at 20°C), water (∼1 g/L at 100°C), and 0.5 N NaOH (0.1 M).
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Keep away from strong oxidizing agents.
RUO – Research Use Only
General References:
- MJ Alowitz; MM Scherer. Kinetics of nitrate, nitrite, and Cr (VI) reduction by iron metal. Environmental Science & Technology.2002, 36 (3), 299-306.
- JB Olmsted; GG Borisy. Ionic and nucleotide requirements for microtubule polymerization in vitro. Biochemistry. 1975, 14, (13), 2996-3005.