Search
Thermo Scientific Chemicals
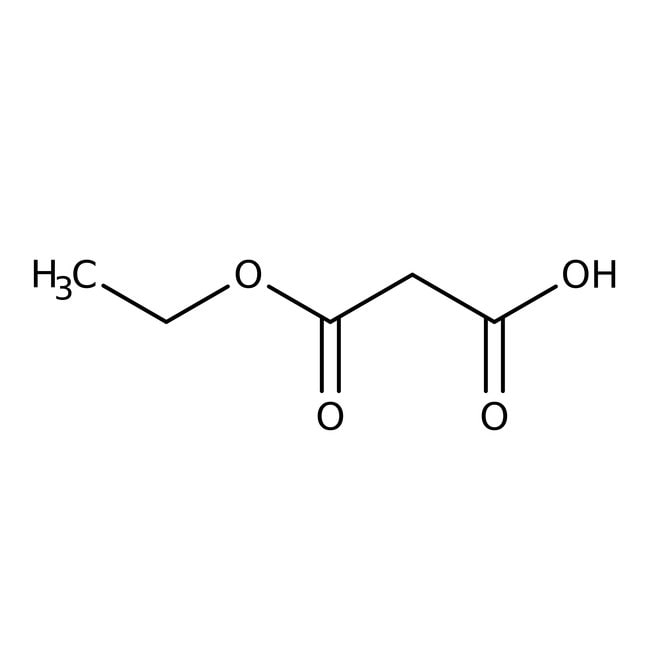
Ethyl hydrogen malonate, 96%
CAS: 1071-46-1 | C5H8O4 | 132.115 g/mol
Catalog number A12627.09
also known as A12627-09
Price (EUR)
45,40
Each
Quantity:
10 g
Price (EUR)
45,40
Each
Chemical Identifiers
CAS1071-46-1
IUPAC Name3-ethoxy-3-oxopropanoic acid
Molecular FormulaC5H8O4
InChI KeyHGINADPHJQTSKN-UHFFFAOYSA-N
SMILESCCOC(=O)CC(O)=O
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Clear colorless to pale yellow
Assay (Aqueous acid-base Titration)≥95.0 to ≤105.0%
Refractive Index1.4245-1.4320 @ 20?C
Water Content (Karl Fischer Titration)≤2%
FormLiquid
Ethyl hydrogen malonate is used as a reactant for the preparation of tetramic acids through Dieckmann ring closure and organocatalytic decarboxylative Doebner-Knoevenagel reactions. It is involved in the acylation reactions and Knoevenagel condensation with aldehydes. It is also used in the preparation of gamma-lactones from olefins by intermolecular carbolactonization in presence of Mn(III) acetate as a catalyst.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Ethyl hydrogen malonate is used as a reactant for the preparation of tetramic acids through Dieckmann ring closure and organocatalytic decarboxylative Doebner-Knoevenagel reactions. It is involved in the acylation reactions and Knoevenagel condensation with aldehydes. It is also used in the preparation of gamma-lactones from olefins by intermolecular carbolactonization in presence of Mn(III) acetate as a catalyst.
Solubility
Miscible with water, chloroform and other solvents.
Notes
Incompatible with strong oxidizing agents.
Ethyl hydrogen malonate is used as a reactant for the preparation of tetramic acids through Dieckmann ring closure and organocatalytic decarboxylative Doebner-Knoevenagel reactions. It is involved in the acylation reactions and Knoevenagel condensation with aldehydes. It is also used in the preparation of gamma-lactones from olefins by intermolecular carbolactonization in presence of Mn(III) acetate as a catalyst.
Solubility
Miscible with water, chloroform and other solvents.
Notes
Incompatible with strong oxidizing agents.
RUO – Research Use Only
General References:
- Reaction with alkyl chloroformates results in decarboxylation of the mixed anhydride in situ, providing a route to mixed ethyl alkyl malonates in high yield under very mild conditions: Tetrahedron Lett., 26, 1573 (1985).
- Half esters of malonic acid react with ɑß-enals in pyridine/ DMAP to give ɑß δ-dienoic esters with good (E)-selectivity: Synthesis: 534 (1988).
- The O,ɑ-dilithio-derivative, formed with n-BuLi in THF, can be acylated with acid chlorides to give, on acidic work-up (in situ decarboxylation), ß-ketoesters directly in high yield: J. Org. Chem., 44, 310 (1979); Org. Synth. Coll., 7, 213 (1990). Similarly, alkylation of the dilithio-derivative provides a direct route to esters of substituted acetic acids: J. Org. Chem., 40, 2556 (1975).
- Carbodiimide coupling with polymer-bound ɑ-amino acids has been used in the solid-phase synthesis of substituted tetramic acids; J. Org. Chem., 63, 4808 (1998).
- See also Ethyl potassium malonate, A10720 .
- Fattorusso, C.; Persico, M.; Basilico, N.; Taramelli, D.; Fattorusso, E.; Scala, F.; Taglialatela-Scafati, O. Antimalarials based on the dioxane scaffold of plakortin. A concise synthesis and SAR studies. Bioorg. Med. Chem. 2011, 19 (1), 312-320.
- Anwar, M.; Moloney, M. G. Chiral Bicyclic Tetramates as Non-Planar Templates for Chemical Library Synthesis. Chem. Biol. Drug Des. 2013, 81 (5), 645-649.