Search
Thermo Scientific Chemicals
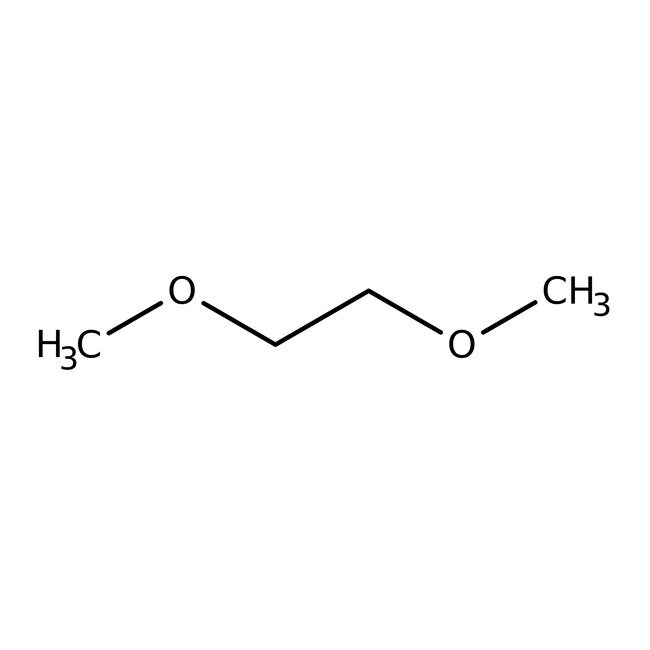
1,2-Dimethoxyethane, 99+%, stab. with BHT
CAS: 110-71-4 | C4H10O2 | 90.122 g/mol
Chemical Identifiers
CAS110-71-4
IUPAC Name1,2-dimethoxyethane
Molecular FormulaC4H10O2
InChI KeyXTHFKEDIFFGKHM-UHFFFAOYSA-N
SMILESCOCCOC
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Clear colorless
Assay (GC)≥99.0%
Identification (FTIR)Conforms
Refractive Index1.3785-1.3815 @ 20?C
FormLiquid
View more
1,2-Dimethoxyethane is widely used as a solvent for electrolyte of lithium batteries, polysilicones, oligo- and polysaccharides. It plays an important role in Grignard reactions, Suzuki reactions and Stille couplings in organometallic chemistry and in pharmaceutical synthesis. It is a higher boiling point solvent and is used as an alternative to diethyl ether and tetrahydrofuran. It is used for the etching of synthetic polymers like polytetrafluoroethylene and other fluoropolymers with alkali metal dispersions.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
1,2-Dimethoxyethane is widely used as a solvent for electrolyte of lithium batteries, polysilicones, oligo- and polysaccharides. It plays an important role in Grignard reactions, Suzuki reactions and Stille couplings in organometallic chemistry and in pharmaceutical synthesis. It is a higher boiling point solvent and is used as an alternative to diethyl ether and tetrahydrofuran. It is used for the etching of synthetic polymers like polytetrafluoroethylene and other fluoropolymers with alkali metal dispersions.
Solubility
Miscible with water, methanol, ethanol, diethyl ether, acetone, tetrahydrofuran, benzene and toluene.
Notes
Keep away from sources of ignition. Keep container tightly closed in a dry and well-ventilated place. Incompatible with strong oxidizing agents. It can be oxidized on air to form peroxides.
1,2-Dimethoxyethane is widely used as a solvent for electrolyte of lithium batteries, polysilicones, oligo- and polysaccharides. It plays an important role in Grignard reactions, Suzuki reactions and Stille couplings in organometallic chemistry and in pharmaceutical synthesis. It is a higher boiling point solvent and is used as an alternative to diethyl ether and tetrahydrofuran. It is used for the etching of synthetic polymers like polytetrafluoroethylene and other fluoropolymers with alkali metal dispersions.
Solubility
Miscible with water, methanol, ethanol, diethyl ether, acetone, tetrahydrofuran, benzene and toluene.
Notes
Keep away from sources of ignition. Keep container tightly closed in a dry and well-ventilated place. Incompatible with strong oxidizing agents. It can be oxidized on air to form peroxides.
RUO – Research Use Only
General References:
- Solvent for lithiations and metal hydride reductions.
- Recommended solvent for the Schmidt reaction of ketones with hydrazoic acid, avoiding potential hazards reported with the more usual chlorinated solvents, particularly chloroform. The hydrazoic acid is generated in situ from NaN3 and methanesulfonic acid: Tetrahedron, 52, 1609 (1996).
- Che, P.; Lu, F.; Si, X.; Xu, J. Catalytic etherification of hydroxyl compounds to methyl ethers with 1,2-dimethoxyethane. RSC Adv. 2015, 5 (31), 24139-24143.
- Monreal, M. J.; Thomson, R. K.; Scott, B. L.; Kiplinger, J. L. Enhancing the synthetic efficacy of thorium tetrachloride bis(1,2-dimethoxyethane) with added 1,2-dimethoxyethane: Preparation of metallocene thorium dichlorides. Inorg. Chem. Commun. 2014, 46, 51-53.

.png-150.jpg)

