Search
Thermo Scientific Chemicals
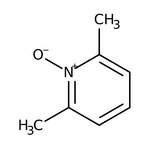
2,6-Lutidine N-oxide, 98%
CAS: 1073-23-0 | C7H9NO | 123.155 g/mol
Catalog number A14299.18
also known as A14299-18
Price (EUR)
147,00
Each
Quantity:
50 g
Price (EUR)
147,00
Each
Chemical Identifiers
CAS1073-23-0
IUPAC Name2,6-dimethylpyridin-1-ium-1-olate
Molecular FormulaC7H9NO
InChI KeyLIDGFHXPUOJZMK-UHFFFAOYSA-N
SMILESCC1=CC=CC(C)=[N+]1[O-]
View more
Specifications Specification Sheet
Specification Sheet
FormLiquid
Refractive Index1.5640-1.5720 @ 20?C
Appearance (Color)Clear colorless to yellow
Assay (GC)≥97.5%
2,6-Lutidine N-oxide is used as a ligand and form coordination complexes with metal salts.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
2,6-Lutidine N-oxide is used as a ligand and form coordination complexes with metal salts.
Solubility
Miscible with water.
Notes
Hygroscopic. Incompatible with oxidizing agents, moisture and water.
2,6-Lutidine N-oxide is used as a ligand and form coordination complexes with metal salts.
Solubility
Miscible with water.
Notes
Hygroscopic. Incompatible with oxidizing agents, moisture and water.
RUO – Research Use Only
General References:
- Yamamoto, K.; Tsurugi, H.; Mashima, K. Direct Evidence for a [4+2] Cycloaddition Mechanism of Alkynes to Tantallacyclopentadiene on Dinuclear Tantalum Complexes as a Model of Alkyne Cyclotrimerization. Chem-Eur. J. 2015, 21 (32), 11369-11377.
- Nösel, P.; dos Santos Comprido, L. N.; Lauterbach, T.; Rudolph, M.; Rominger, F.; Hashmi, A. S. K. 1,6-Carbene Transfer: Gold-Catalyzed Oxidative Diyne Cyclizations. J. Am. Chem. Soc. 2013, 135 (41), 15662-15666.