Search
Thermo Scientific Chemicals
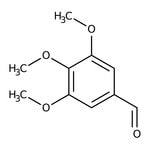
3,4,5-Trimethoxybenzaldehyde, 98%
CAS: 86-81-7 | C10H12O4 | 196.202 g/mol
Catalog number A15038.14
also known as A15038-14
Price (EUR)
24,80
Each
Quantity:
25 g
Price (EUR)
24,80
Each
Chemical Identifiers
CAS86-81-7
IUPAC Name3,4,5-trimethoxybenzaldehyde
Molecular FormulaC10H12O4
InChI KeyOPHQOIGEOHXOGX-UHFFFAOYSA-N
SMILESCOC1=CC(C=O)=CC(OC)=C1OC
View more
Specifications Specification Sheet
Specification Sheet
Assay (GC)≥97.5%
Free acid (titration)≤1%
Identification (FTIR)Conforms
Appearance (Color)White to cream to pale orange
Melting Point (clear melt)72.0-78.0?C
View more
3,4,5-Trimethoxybenzaldehyde is used in the synthesis of trimethoprim and psychedelic phenethylamines. Also it can also be used in the production of plastic additives.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
3,4,5-Trimethoxybenzaldehyde is used in the synthesis of trimethoprim and psychedelic phenethylamines. Also it can also be used in the production of plastic additives.
Solubility
Soluble in water (1.49 mg/L at 25°C), alcohol, toluene, acetone, and methanol.
Notes
Air Sensitive, store away from air. Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Store away from oxidizing agent.
3,4,5-Trimethoxybenzaldehyde is used in the synthesis of trimethoprim and psychedelic phenethylamines. Also it can also be used in the production of plastic additives.
Solubility
Soluble in water (1.49 mg/L at 25°C), alcohol, toluene, acetone, and methanol.
Notes
Air Sensitive, store away from air. Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Store away from oxidizing agent.
RUO – Research Use Only
General References:
- D.V Rao: F.A. Stuber. An efficient synthesis of 3, 4, 5-trimethoxybenzaldehyde from vanillin. Synthesis, 2013, 4 680-689.
- Percy S. Manchand; Peter S. Belica; Harry S. Wonga. Synthesis of 3,4,5-Trimethoxybenzaldehyde. Talanta. 1990, 20(17),2659-2666.