Search
Thermo Scientific Chemicals
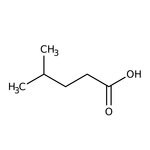
4-Methylvaleric acid, 99%
CAS: 646-07-1 | C6H12O2 | 116.16 g/mol
Catalog number A15405.22
also known as A15405-22
Price (EUR)
154,00
Each
Quantity:
100 g
Price (EUR)
154,00
Each
Chemical Identifiers
CAS646-07-1
IUPAC Name4-methylpentanoic acid
Molecular FormulaC6H12O2
InChI KeyFGKJLKRYENPLQH-UHFFFAOYSA-N
SMILESCC(C)CCC(O)=O
View more
Specifications Specification Sheet
Specification Sheet
Refractive Index1.4125-1.4155 @ 20?C
Assay (Aqueous acid-base Titration)≥98.5 to ≤101.5%
FormLiquid
Identification (FTIR)Conforms
Appearance (Color)Clear colorless
View more
4-Methylvaleric acid has been used in asymmetric synthesis of (4S)-4-isopropyl-3-[(2?S)-2?,4?-dimethyl-valeryl)]-2-oxazolidinone. It is a versatile building block used in the synthesis of various pharmaceutical and biologically active compounds, including inhibitors and antagonists. It is involved in the synthesis of novel series of high affinity, functionally potent antagonists of the CCR1 receptor.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
4-Methylvaleric acid has been used in asymmetric synthesis of (4S)-4-isopropyl-3-[(2′S)-2′,4′-dimethyl-valeryl)]-2-oxazolidinone. It is a versatile building block used in the synthesis of various pharmaceutical and biologically active compounds, including inhibitors and antagonists. It is involved in the synthesis of novel series of high affinity, functionally potent antagonists of the CCR1 receptor.
Solubility
Soluble in alcohol. Slightly soluble in water.
Notes
Store in cool, dry conditions in a well sealed container. Store away from oxidizing agents, bases.
4-Methylvaleric acid has been used in asymmetric synthesis of (4S)-4-isopropyl-3-[(2′S)-2′,4′-dimethyl-valeryl)]-2-oxazolidinone. It is a versatile building block used in the synthesis of various pharmaceutical and biologically active compounds, including inhibitors and antagonists. It is involved in the synthesis of novel series of high affinity, functionally potent antagonists of the CCR1 receptor.
Solubility
Soluble in alcohol. Slightly soluble in water.
Notes
Store in cool, dry conditions in a well sealed container. Store away from oxidizing agents, bases.
RUO – Research Use Only
General References:
- Kazutoku Ohta; Atsuya Towata; Masayoshi Ohashi; Toyohide Takeuchi. Separation and conductimetric detection of C1-C7 aliphatic monocarboxylic acids and C1-C7 aliphatic monoamines on unfunctionized polymethacrylate resin columns. Journal of Chromatography A. 2004, 1039, (1-2), 171-177.
- Vincent Guerlavais; Patrick J. Carroll; Madeleine M. Joullié. Progress towards the total synthesis of callipeltin A. Asymmetric synthesis of (2R, 3R, 4S)-3-hydroxy-2, 4, 6-trimethylheptanoic acid. Tetrahedron: Asymmetry. 2002, 13(7), 675-680.