Search
Thermo Scientific Chemicals
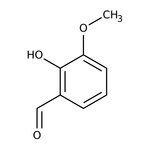
2-Hydroxy-3-methoxybenzaldehyde, 99%
CAS: 148-53-8 | C8H8O3 | 152.15 g/mol
Catalog number A15672.22
also known as A15672-22
Price (EUR)
53,40
Each
Quantity:
100 g
Price (EUR)
53,40
Each
Chemical Identifiers
CAS148-53-8
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Yellow to brown
FormCrystals or powder or crystalline powder or flakes
Assay (GC)≥98.5%
Identification (FTIR)Conforms
Melting Point (clear melt)40-45°C
o-Vanillin has been used to study the solvent-free reaction between o-vanillin and p-toluidine using NMR, DSC and XRD analysis. It was used in the synthesis of new ligand for Fe(III) and Al(lII). It is also used in the study of mutagenesis and as a synthetic precursor for pharmaceuticals.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
o-Vanillin has been used to study the solvent-free reaction between o-vanillin and p-toluidine using NMR, DSC and XRD analysis. It was used in the synthesis of new ligand for Fe(III) and Al(lII). It is also used in the study of mutagenesis and as a synthetic precursor for pharmaceuticals.
Solubility
Solubility in methanol is almost transparent. Soluble in alcohol, methanol and dioxane (1g in 10 mL). Insoluble in water.
Notes
Air sensitive. Store away from air and oxidizing agents. Keep the container tightly closed and place it in a cool, dry and well ventilated condition.
o-Vanillin has been used to study the solvent-free reaction between o-vanillin and p-toluidine using NMR, DSC and XRD analysis. It was used in the synthesis of new ligand for Fe(III) and Al(lII). It is also used in the study of mutagenesis and as a synthetic precursor for pharmaceuticals.
Solubility
Solubility in methanol is almost transparent. Soluble in alcohol, methanol and dioxane (1g in 10 mL). Insoluble in water.
Notes
Air sensitive. Store away from air and oxidizing agents. Keep the container tightly closed and place it in a cool, dry and well ventilated condition.
RUO – Research Use Only
General References:
- Dolotko O, et al. Mechanically induced reactions in organic solids: liquid eutectics or solid-state processes?.New J. Chem.,2010,34(1), 25-28.
- Valeria M Nurchi, et al. Kojic acid derivatives as powerful chelators for iron(III) and aluminum(III).Dalton Trans.,2011,40(22), 5984-5998.