Search
Thermo Scientific Chemicals
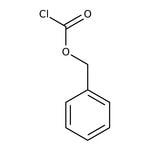
Benzyl chloroformate, 95%, stab. with ca 0.1% sodium carbonate
CAS: 501-53-1 | C8H7ClO2 | 170.592 g/mol
Price (EUR)
32,30
Each
Chemical Identifiers
CAS501-53-1
IUPAC Namebenzyl carbonochloridate
Molecular FormulaC8H7ClO2
InChI KeyHSDAJNMJOMSNEV-UHFFFAOYSA-N
SMILESClC(=O)OCC1=CC=CC=C1
View more
Specifications Specification Sheet
Specification Sheet
Refractive Index1.5140-1.5200 @ 20?C
Identification (FTIR)Conforms
Appearance (Color)Clear colorless to pale yellow
Assay (GC)≥94.0%
FormLiquid
Benzyl chloroformate is widely used as a reactive chemical intermediate in plastic, pharmaceutical, agricultural and organic chemicals. It is useful for the introduction of carboxybenzyl (cbz) protecting group for amines such as aniline in organic synthesis. It is also involved in the synthesis of 1,2,4-oxadiazoles.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Benzyl chloroformate is widely used as a reactive chemical intermediate in plastic, pharmaceutical, agricultural and organic chemicals. It is useful for the introduction of carboxybenzyl (cbz) protecting group for amines such as aniline in organic synthesis. It is also involved in the synthesis of 1,2,4-oxadiazoles.
Solubility
Miscible with ether, acetone and benzene.
Notes
Store in cool place. Moisture sensitive.
Benzyl chloroformate is widely used as a reactive chemical intermediate in plastic, pharmaceutical, agricultural and organic chemicals. It is useful for the introduction of carboxybenzyl (cbz) protecting group for amines such as aniline in organic synthesis. It is also involved in the synthesis of 1,2,4-oxadiazoles.
Solubility
Miscible with ether, acetone and benzene.
Notes
Store in cool place. Moisture sensitive.
RUO – Research Use Only
General References:
- Kim, J. S.; Kang, J. C.; Yoo, G. H.; Jin, X.; Myeong, I. S.; Oh, C. Y.; Ham, W. H. Stereoselective total synthesis of (-)-conduramine F-1 via chiral 1,3-oxazine. Tetrahedron 2015, 71 (18), 2772-2776.
- Lezama, J.; Domínguez, R. M.; Chuchani, G. Kinetics of the Gas-Phase Elimination Reaction of Benzyl Chloroformate and Neopentyl Chloroformate. Int. J. Chem. Kinet. 2015, 47 (2), 104-112.