Search
Thermo Scientific Chemicals
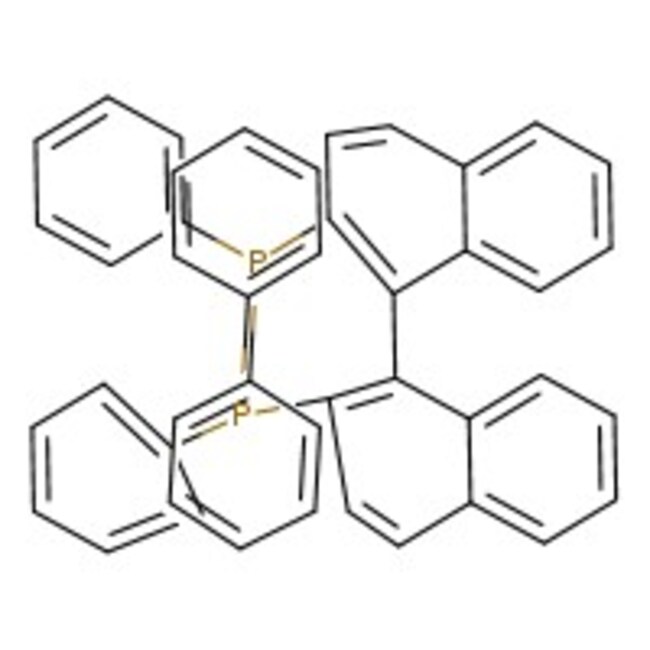
(+/-)-2,2'-Bis(diphenylphosphino)-1,1'-binaphthyl, 97+%
CAS: 98327-87-8 | C44H32P2 | 622.69 g/mol
Catalog number H26226.14
also known as H26226-14
Price (EUR)
454,00
Each
Quantity:
25 g
Price (EUR)
454,00
Each
Chemical Identifiers
CAS98327-87-8
IUPAC Name[2'-(diphenylphosphanyl)-[1,1'-binaphthalen]-2-yl]diphenylphosphane
Molecular FormulaC44H32P2
InChI KeyMUALRAIOVNYAIW-UHFFFAOYSA-N
SMILESC1=CC=C(C=C1)P(C1=CC=CC=C1)C1=C(C2=CC=CC=C2C=C1)C1=C(C=CC2=CC=CC=C12)P(C1=CC=CC=C1)C1=CC=CC=C1
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)White to pale cream
FormPowder
Phosphorus-31 NMRConforms to structure
Purity (DSC)≥97.0%
Identification (FTIR)Conforms
View more
(±)-2,2'-Bis(diphenylphosphino)-1,1'-binaphthyl plays an essential role in the organic synthesis of enantioselective transformations catalyzed by the complexes of ruthenium, rhodium and palladium. It is also employed in palladium-catalyzed arylamine coupling in the preparation of demethylthiocholchines. It is used with Cu(II) to catalyze the addition of arylsulfonamides to styrenes and olefins.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
(^+)-2,2′-Bis(diphenylphosphino)-1,1′-binaphthyl plays an essential role in the organic synthesis of enantioselective transformations catalyzed by the complexes of ruthenium, rhodium and palladium. It is also employed in palladium-catalyzed arylamine coupling in the preparation of demethylthiocholchines. It is used with Cu(II) to catalyze the addition of arylsulfonamides to styrenes and olefins.
Solubility
Soluble in tetrahydrofuran, benzene and dichloromethane. Slightly soluble in ether, methanol and ethanol. Insoluble in water.
Notes
Air sensitive. Keep container tightly closed in a dry and well-ventilated place.
(^+)-2,2′-Bis(diphenylphosphino)-1,1′-binaphthyl plays an essential role in the organic synthesis of enantioselective transformations catalyzed by the complexes of ruthenium, rhodium and palladium. It is also employed in palladium-catalyzed arylamine coupling in the preparation of demethylthiocholchines. It is used with Cu(II) to catalyze the addition of arylsulfonamides to styrenes and olefins.
Solubility
Soluble in tetrahydrofuran, benzene and dichloromethane. Slightly soluble in ether, methanol and ethanol. Insoluble in water.
Notes
Air sensitive. Keep container tightly closed in a dry and well-ventilated place.
RUO – Research Use Only
General References:
- Hatano, B.; Hashimoto, K.; Katagiri, H.; Kijima, T.; Murakami, S.; Matsuba, S.; Kusakari, M. Enantioresolution of 2,2'-Bis(diphenylphosphino)-1,1'-binaphthyl Oxide Using Inclusion Complex with Chiral 2,2'-Dihydroxy-1,1'-binaphtyl. J. Org. Chem. 2012, 77 (7), 3595-3597.
- Deng, C.; Ou, G.; She, J.; Yuan, Y. Biphasic asymmetric hydroformylation and hydrogenation by water-soluble rhodium and ruthenium complexes of sulfonated (R)-2,2'-bis(diphenylphosphino)-1,1'-binaphthyl in ionic liquids. J. Mol. Catal. A: Chem. 2007, 270 (1-2), 76-82.
- Atesin, A. C.; Zhang, J.; Vaidya, T.; Brennessel, W. W.; Frontier, A. J.; Eisenberg, R. Synthesis, Characterization, and Catalytic Properties of New Electrophilic Iridium(III) Complexes Containing the (R)-(+)-2,2'-Bis(diphenylphosphino)-1,1'-binaphthyl Ligand. Inorg. Chem. 2010, 49 (9), 4331-4342.