Search
Thermo Scientific Chemicals
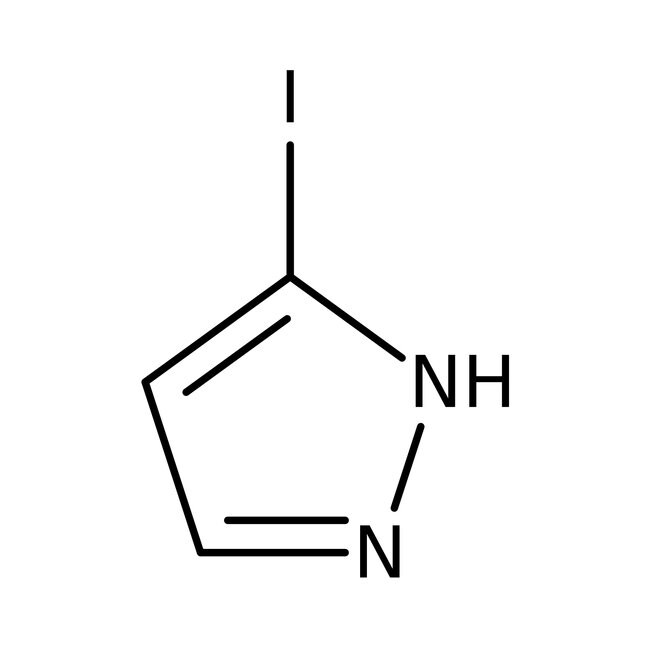
3-Iodo-1H-pyrazole, 97%
CAS: 4522-35-4 | C3H3IN2 | 193.98 g/mol
Catalog number H36465.06
also known as H36465-06
Price (EUR)
186,00
Each
Quantity:
5 g
Price (EUR)
186,00
Each
Chemical Identifiers
CAS4522-35-4
IUPAC Name5-iodo-1H-pyrazole
Molecular FormulaC3H3IN2
InChI KeyRUKDVLFJSMVBLV-UHFFFAOYSA-N
SMILESIC1=CC=NN1
View more
Specifications Specification Sheet
Specification Sheet
FormPowder
Appearance (Color)Yellow
Assay (GC)≥96.0%
Melting Point (clear melt)66.0-75.0?C
3-Iodo-1H-pyrazole is used as pharmaceutical intermediate.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
3-Iodo-1H-pyrazole is used as pharmaceutical intermediate.
Solubility
Insoluble in water.
Notes
Light sensitive. Incompatible with oxidizing agents, acids and bases.
3-Iodo-1H-pyrazole is used as pharmaceutical intermediate.
Solubility
Insoluble in water.
Notes
Light sensitive. Incompatible with oxidizing agents, acids and bases.
RUO – Research Use Only
General References:
- Rita Mazeikaite; Jurgis Sudzius; Gintaras Urbelis; Linas Labanauskas. Synthesis of substituted-3-iodo-1H-pyrazole derivatives and their further modification under Sonogashira cross-coupling reaction conditions. ARKIVOC. 2014, 6, 54-71
- Taiki Morita; Daisuke Kobayashi; Keisuke Matsumura; Dr. Kohei Johmoto; Dr. Hidehiro Uekusa; Dr. Shinichiro Fuse; Prof. Dr. Takashi Takahash. Sequential SNAr reaction/Suzuki-Miyaura Coupling/C-H direct arylations approach for the rapid synthesis of tetraaryl-substituted pyrazoles. Chemistry - An Asian Journal. 2015, 10, (8),1626-1630