Search
Thermo Scientific Chemicals
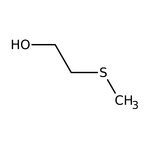
2-(Methylthio)ethanol, 99%
CAS: 5271-38-5 | C3H8OS | 92.16 g/mol
Catalog number L05442.14
also known as L05442-14
Price (EUR)
53,40
Each
Quantity:
25 g
Price (EUR)
53,40
Each
Chemical Identifiers
CAS5271-38-5
IUPAC Name2-(methylsulfanyl)ethan-1-ol
Molecular FormulaC3H8OS
InChI KeyWBBPRCNXBQTYLF-UHFFFAOYSA-N
SMILESCSCCO
View more
Specifications Specification Sheet
Specification Sheet
FormLiquid
Appearance (Color)Clear colorless to pale yellow
Refractive Index1.4930-1.4960 @ 20?C
Assay (GC)>98.5%
2-(Methylthio)ethanol is used as an important raw material and intermediate used in organic synthesis, pharmaceuticals, agrochemicals and dyestuffs.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
2-(Methylthio)ethanol is used as an important raw material and intermediate used in organic synthesis, pharmaceuticals, agrochemicals and dyestuffs.
Solubility
Not miscible in water.
Notes
Do not store together with acids. Store away from Oxidizing agents.
2-(Methylthio)ethanol is used as an important raw material and intermediate used in organic synthesis, pharmaceuticals, agrochemicals and dyestuffs.
Solubility
Not miscible in water.
Notes
Do not store together with acids. Store away from Oxidizing agents.
RUO – Research Use Only
General References:
- Michael L. McKee. Comparison of Gas-Phase and Solution-Phase Reactions of Dimethyl Sulfide and 2-(Methylthio)ethanol with Hydroxyl Radical.J. Phys. Chem. A.2003,107 (35), 6819-6827.
- Reagent for carboxyl group protection in peptide synthesis. Cleavage is by alkylation (MeI), followed by mild alkaline hydrolysis: J. Chem. Soc. (C), 807 (1966); Chem. Ber., 109, 3693 (1976). See Appendix 6.