Search
Thermo Scientific Chemicals
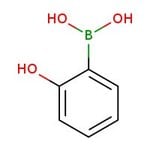
2-Hydroxybenzeneboronic acid, 97%
CAS: 89466-08-0 | C6H7BO3 | 137.93 g/mol
Catalog number L19400.06
also known as L19400-06
Price (EUR)
129,00
Each
Quantity:
5 g
Price (EUR)
129,00
Each
Chemical Identifiers
CAS89466-08-0
IUPAC Name(2-hydroxyphenyl)boronic acid
Molecular FormulaC6H7BO3
InChI KeyYDMRDHQUQIVWBE-UHFFFAOYSA-N
SMILESOB(O)C1=CC=CC=C1O
View more
Specifications Specification Sheet
Specification Sheet
FormPowder
Assay (HPLC)≥96.0%
Appearance (Color)White to pale cream
Assay (Aqueous acid-base Titration)≥96.0%
Proton NMRConforms to structure
2-Hydroxybenzeneboronic acid is used as a pharmaceutical intermediate.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
2-Hydroxybenzeneboronic acid is used as a pharmaceutical intermediate.
Solubility
Insoluble in water.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Incompatible with oxidizing agents.
2-Hydroxybenzeneboronic acid is used as a pharmaceutical intermediate.
Solubility
Insoluble in water.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Incompatible with oxidizing agents.
RUO – Research Use Only
General References:
- Takeo Konakahara.; Y.B. Kiran.; Yuri Okuno.; Reiko Ikeda.; Norio Sakai. An expedient synthesis of ellipticine via Suzuki-Miyaura coupling. Tetrahedron Letters. 2010, 51 (17), 2335-2338.
- Alexandre Requet.; Amel Souibgui.; Grégory Pieters.; Sabrina Ferhi.; Alicia Letaieff.; Abel Carlin-Sinclair.; Sylvain Marque.; Jérome Marrot.; Béchir Ben Hassine.; Anne Gaucher.; Damien Prim. Synthesis of partially hydrogenated oxa[5] and oxa[6]helicenes from β-chlorovinylaldehydes. Tetrahedron Letters. 2013, 55 (35), 4721-4725.