Search
Thermo Scientific Chemicals
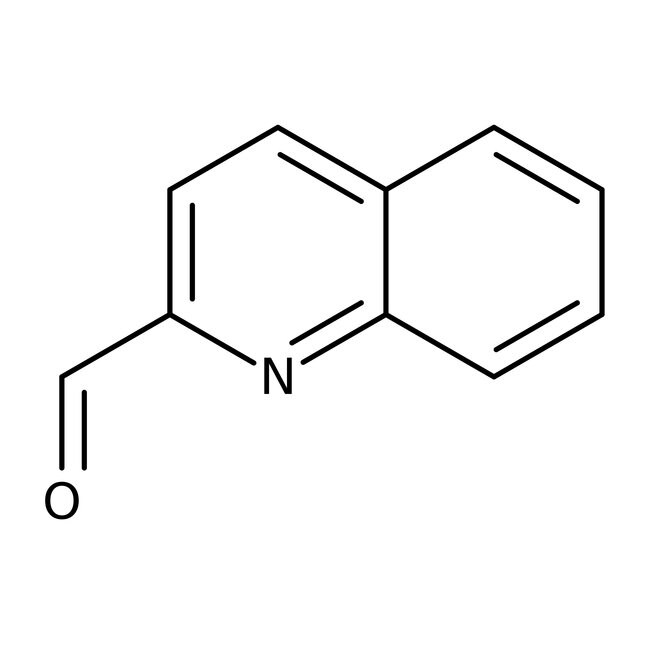
Quinoline-2-carboxaldehyde, 97%
CAS: 5470-96-2 | C10H7NO | 157.17 g/mol
Catalog number L20202.06
also known as L20202-06
Price (EUR)
73,20
Each
Quantity:
5 g
Price (EUR)
73,20
Each
Chemical Identifiers
CAS5470-96-2
Specifications Specification Sheet
Specification Sheet
FormCrystals or powder or crystalline powder
Free acid (titration)≤1.5%
Assay (GC)≥96.0%
Melting Point64.0-70.0?C
Appearance (Color)Yellow to orange to brown
2-Quinolinecarboxaldehyde was used in the preparation of 3-(2-quinolyl)-1-phenyl-2-propenone via rapid, tandem aldol-Michael reactions with the lithium, sodium and potassium enolates of acetophenone; imine-type ligands; sugar-quinoline fluorescent sensor for the detection of Hg2+ in natural water.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
2-Quinolinecarboxaldehyde was used in the preparation of 3-(2-quinolyl)-1-phenyl-2-propenone via rapid, tandem aldol-Michael reactions with the lithium, sodium and potassium enolates of acetophenone; imine-type ligands; sugar-quinoline fluorescent sensor for the detection of Hg2+ in natural water.
Solubility
Insoluble in water.
Notes
Stable under recommended storage conditions. Incompatible with oxidizing agents.
2-Quinolinecarboxaldehyde was used in the preparation of 3-(2-quinolyl)-1-phenyl-2-propenone via rapid, tandem aldol-Michael reactions with the lithium, sodium and potassium enolates of acetophenone; imine-type ligands; sugar-quinoline fluorescent sensor for the detection of Hg2+ in natural water.
Solubility
Insoluble in water.
Notes
Stable under recommended storage conditions. Incompatible with oxidizing agents.
RUO – Research Use Only
General References:
- Wachter-Jurcsak N, et al. Addressing the unusual reactivity of 2-pyridinecarboxaldehyde and 2-quinolinecarboxaldehyde in base-catalyzed aldol reactions with acetophenone. Tetrahedron Lett.1998,39(23), 3903-6
- Attilio Ardizzoia G, et al. Copper (I)-imine complexes: Synthesis and catalytic activity in olefin cyclopropanation. Inorganica Chim. Acta.2009,362(10), 3507-12