Search
Thermo Scientific Chemicals
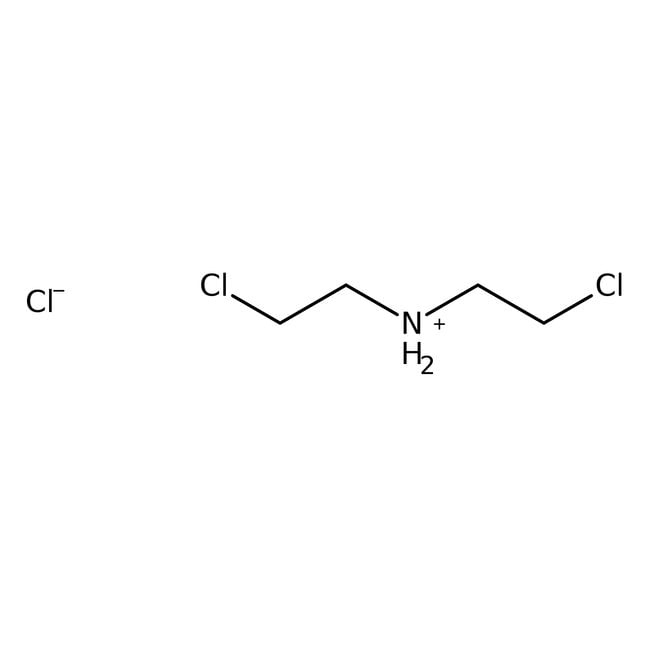
Bis(2-chloroethyl)amine hydrochloride, 98%
CAS: 821-48-7 | C4H10Cl3N | 178.481 g/mol
Catalog number A13118.18
also known as A13118-18
Price (EUR)
21,10
Each
Quantity:
50 g
Price (EUR)
21,10
Each
Chemical Identifiers
CAS821-48-7
IUPAC Namebis(2-chloroethyl)azanium chloride
Molecular FormulaC4H10Cl3N
InChI KeyYMDZDFSUDFLGMX-UHFFFAOYSA-N
SMILES[Cl-].ClCC[NH2+]CCCl
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)White to cream to pale brown
FormCrystals or powder or crystalline powder or flakes
Assay (Titration ex Chloride)≥97.5 to ≤102.5%
Identification (FTIR)Conforms
Melting Point (clear melt)212.0-220.0?C
Bis(2-chloroethyl)amine hydrochloride is used as an intermediate in chemicals synthesis and building block for piperazine derivatives.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Bis(2-chloroethyl)amine hydrochloride is used as an intermediate in chemicals synthesis and building block for piperazine derivatives.
Solubility
Partly miscible in water.
Notes
Stable under recommended storage conditions. Incompatible with oxidizing agents and bases.
Bis(2-chloroethyl)amine hydrochloride is used as an intermediate in chemicals synthesis and building block for piperazine derivatives.
Solubility
Partly miscible in water.
Notes
Stable under recommended storage conditions. Incompatible with oxidizing agents and bases.
RUO – Research Use Only
General References:
- Daan Noort; Albert G.Hulst; Rob Jansen. Covalent binding of nitrogen mustards to the cysteine-34 residue in human serum albumin. Archives of Toxicology. 2002, 76, (2),83-88
- G.Momerency; K.Van Cauwenberghe; P.H.Slee; A.T.Van Oosterom; E.A De Bruijn. The determination of cyclophosphamide and its metabolites in blood plasma as stable trifluoroacetyl derivatives by electron capture chemical ionization gas chromatography/mass spectrometry. Biological Mass Spectrometry. 1994, 23, (3),149-158