Search
Thermo Scientific Chemicals
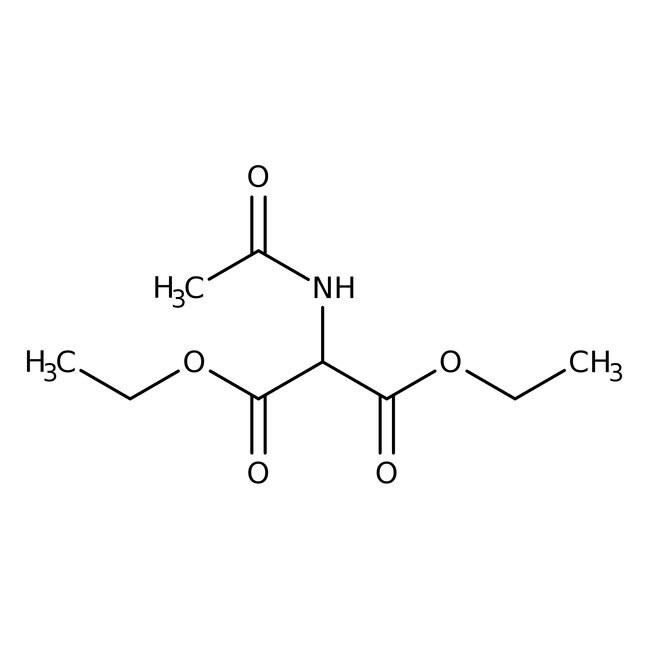
Diethyl acetamidomalonate, 98+%
CAS: 1068-90-2 | C9H15NO5 | 217.221 g/mol
Catalog number A14048.36
also known as A14048-36
Price (EUR)
152,00
Each
Quantity:
500 g
Price (EUR)
152,00
Each
Chemical Identifiers
CAS1068-90-2
IUPAC Name1,3-diethyl 2-acetamidopropanedioate
Molecular FormulaC9H15NO5
InChI KeyISOLMABRZPQKOV-UHFFFAOYSA-N
SMILESCCOC(=O)C(NC(C)=O)C(=O)OCC
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)White
FormCrystals or powder or crystalline powder or fused solid
Assay (GC)≥98.0%
Identification (FTIR)Conforms
Melting Point (clear melt)94.0-100.0?C
Diethyl 2-Acetamidomalonate, is a versatile building block used for the synthesis of various pharmaceutical and biologically active compounds. It is an intermediate for the preparation of Novobiocin analogues as potential heat shock protein 90 inhibitors. It is also used as a important intermediates in syntheses of vitamins B1 and B6, barbiturates, non-steroidal anti-inflammatory agents, other numerous pharmaceuticals.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Diethyl 2-Acetamidomalonate, is a versatile building block used for the synthesis of various pharmaceutical and biologically active compounds. It is an intermediate for the preparation of Novobiocin analogues as potential heat shock protein 90 inhibitors. It is also used as a important intermediates in syntheses of vitamins B1 and B6, barbiturates, non-steroidal anti-inflammatory agents, other numerous pharmaceuticals.
Solubility
Soluble in chloroform and methanol. Slightly soluble in water.
Notes
Store in cool, dry place in tightly closed containers. Store away from oxidizing agent.
Diethyl 2-Acetamidomalonate, is a versatile building block used for the synthesis of various pharmaceutical and biologically active compounds. It is an intermediate for the preparation of Novobiocin analogues as potential heat shock protein 90 inhibitors. It is also used as a important intermediates in syntheses of vitamins B1 and B6, barbiturates, non-steroidal anti-inflammatory agents, other numerous pharmaceuticals.
Solubility
Soluble in chloroform and methanol. Slightly soluble in water.
Notes
Store in cool, dry place in tightly closed containers. Store away from oxidizing agent.
RUO – Research Use Only
General References:
- Sambasivarao Kotha; Kuldeep Singh. N-Alkylation of diethyl acetamidomalonate: synthesis of constrained amino acid derivatives by ring-closing metathesis. Tetrahedron Letters. 2004, 45(52), 9607-9610.
- Paul Angell; Peter G. Blazecka; Mark Lovdahl; and Ji Zhang. Indium(III)-Catalyzed Addition of Diethyl Acetamidomalonate to Terminal Alkynes: An Efficient Approach to β-Branched α-Amino Acids. J. Org. Chem. 2007, 72(17), 6606-6609.
- Intermediate for synthesis of racemic amino acids, by reaction with an electrophile, e.g alkyl halide, followed by hydrolysis and decarboxylation. The reactivity can be reversed by conversion to the dehydro-derivative, which then undergoes nucleophilic attack with alkyllithiums, Grignards, malonate anion, etc.: Angew. Chem. Int. Ed., 21, 203 (1982):
- Substituents can also be introduced by Michael addition to a wide variety of acceptors: Synth. Commun., 18, 367 (1988).
- For use in a novel pyrrole synthesis, see 1,4-Dichloro-2-butyne, A13300.
- Compare also Diethyl formamidomalonate, L01189.