Search
Thermo Scientific Chemicals
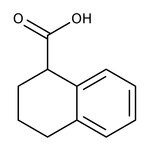
1,2,3,4-Tetrahydro-1-naphthoic acid, 98%
CAS: 1914-65-4 | C11H12O2 | 176.215 g/mol
Catalog number H32138.MD
also known as H32138-MD
Price (EUR)
83,60
Each
Quantity:
250 mg
Price (EUR)
83,60
Each
Chemical Identifiers
CAS1914-65-4
IUPAC Name1,2,3,4-tetrahydronaphthalene-1-carboxylic acid
Molecular FormulaC11H12O2
InChI KeyVDLWTJCSPSUGOA-UHFFFAOYNA-N
SMILESOC(=O)C1CCCC2=CC=CC=C12
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)White to pale cream
FormPowder
Assay (Aqueous acid-base Titration)≥97.5 to ≤102.5%
Assay (Silylated GC)≥97.5%
Melting Point (clear melt)79.0-86.0°C
1,2,3,4-Tetrahydro-1-naphthoic acid is used as an important raw material and intermediate used in organic Synthesis, pharmaceuticals, agrochemicals and dyestuff. 1,2,3,4-tetrahydronaphthalene-1-carboxylic acid is a reagent used to produce protease inhibitors.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
1,2,3,4-Tetrahydro-1-naphthoic acid is used as an important raw material and intermediate used in organic Synthesis, pharmaceuticals, agrochemicals and dyestuff. 1,2,3,4-tetrahydronaphthalene-1-carboxylic acid is a reagent used to produce protease inhibitors.
Solubility
Soluble in methanol, and chloroform.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Stable under recommended storage conditions. Keep away from strong oxidizing agents.
1,2,3,4-Tetrahydro-1-naphthoic acid is used as an important raw material and intermediate used in organic Synthesis, pharmaceuticals, agrochemicals and dyestuff. 1,2,3,4-tetrahydronaphthalene-1-carboxylic acid is a reagent used to produce protease inhibitors.
Solubility
Soluble in methanol, and chloroform.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Stable under recommended storage conditions. Keep away from strong oxidizing agents.
RUO – Research Use Only
General References:
- WA Jacobs.; RG Gould. The ergot alkaloids xii. the synthesis of substances related to lysergic acid. Journal of Biological Chemistry. 1937115 (227), 141-145.
- B Feringa.; H Wynberg. Asymmetric phenol oxidation. Stereospecific and stereoselective oxidative coupling of a chiral tetrahydronaphthol. The Journal of Organic Chemistry. 198146 (12), 2547-2557.