Search
Thermo Scientific Chemicals
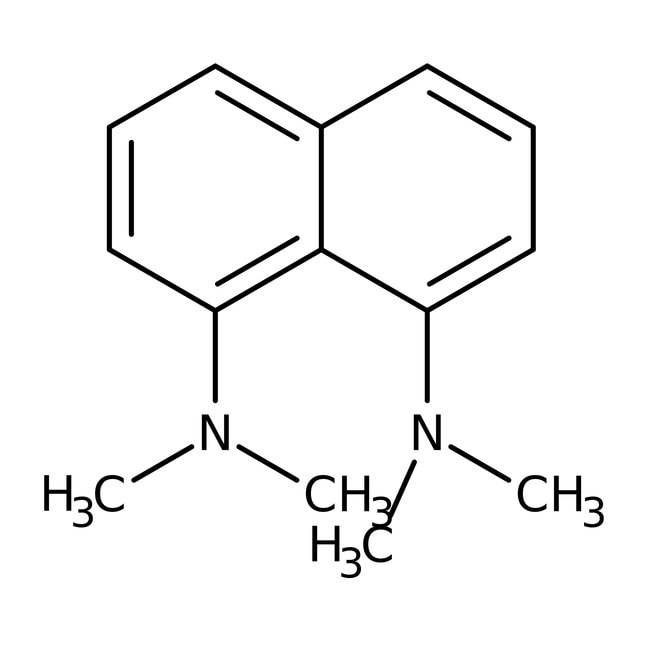
1,8-Bis(dimethylamino)naphthalene, 98+%
CAS: 20734-58-1 | C14H18N2 | 214.312 g/mol
Catalog number L00313.18
also known as L00313-18
Price (EUR)
150,00
Each
Quantity:
50 g
Price (EUR)
150,00
Each
Chemical Identifiers
CAS20734-58-1
IUPAC NameN1,N1,N8,N8-tetramethylnaphthalene-1,8-diamine
Molecular FormulaC14H18N2
InChI KeyGJFNRSDCSTVPCJ-UHFFFAOYSA-N
SMILESCN(C)C1=CC=CC2=CC=CC(N(C)C)=C12
View more
Specifications Specification Sheet
Specification Sheet
FormCrystalline powder
Melting Point (clear melt)46-52?C
Appearance (Color)White to cream to yellow to pink or pink-brown
Assay (GC)≥98.0%
1,8-Bis(dimethylamino)naphthalene is used in the synthesis of C-substituted t-BuNH-8,9-R,R?-nido-7,8,9-C3B8H9 (R,R? = H,H; MeH; Me,Me; Ph,H and Ph,Ph) tricarbollide compounds. It was also used in the preparation of saturated fluoroalkyl(hydrido) complexes of Iridium.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
1,8-Bis(dimethylamino)naphthalene is used in the synthesis of C-substituted t-BuNH-8,9-R,R′-nido-7,8,9-C3B8H9 (R,R′ = H,H; MeH; Me,Me; Ph,H and Ph,Ph) tricarbollide compounds. It was also used in the preparation of saturated fluoroalkyl(hydrido) complexes of Iridium.
Solubility
Soluble in organic solvents.
Notes
Air & Light Sensitive. Keep container tightly closed. Store in cool, dry conditions in well sealed containers.
1,8-Bis(dimethylamino)naphthalene is used in the synthesis of C-substituted t-BuNH-8,9-R,R′-nido-7,8,9-C3B8H9 (R,R′ = H,H; MeH; Me,Me; Ph,H and Ph,Ph) tricarbollide compounds. It was also used in the preparation of saturated fluoroalkyl(hydrido) complexes of Iridium.
Solubility
Soluble in organic solvents.
Notes
Air & Light Sensitive. Keep container tightly closed. Store in cool, dry conditions in well sealed containers.
RUO – Research Use Only
General References:
- Fumiyuki Ozawa.; Akihiko Kubo.; Tamio Hayashi. Palladium-catalyzed asymmetric arylation of 2,3-dihydrofuran: 1,8-Bis(dimethylamino)naphthalene as an efficient base. Tetrahedron Letters. 1992, 33, (11), 1485-1488.
- Danuta Pyzalska.; Robert Pyzalski.; Teresa Borowiak. Structure of 1,8-bis(dimethylamino)naphthalene hydrobromide dihydrate. Journal of Crystallographic and Spectroscopic Research. 1983, 13, (3), 211-220.
- The steric strain is relieved on protonation, hence it is one of the strongest organic bases known, but is only weakly nucleophilic: Chem. Commun., 723 (1968). In DMF vic-dibromides are debrominated in high yield: Synth. Commun., 5, 87 (1975).
- Has been used In combination with Diphenyl phosphonic azide, A12124, for the conversion of carboxylic acids to isocyanates: Synth. Commun., 23, 335 (1993). Preferred base for the conversion of aliphatic amines to isocyanates with Trichloromethyl chloroformate, A17444.