Search
Thermo Scientific Chemicals
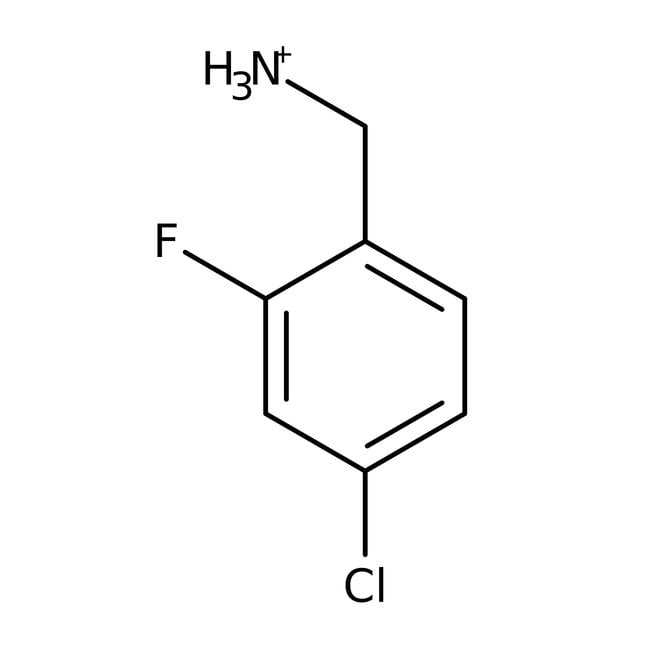
4-Chloro-2-fluorobenzylamine hydrochloride, 97%
CAS: 202982-63-6 | C7H8Cl2FN | 196.046 g/mol
Catalog number L13488.03
also known as L13488-03
Price (EUR)
23,20
Each
Quantity:
1 g
Price (EUR)
23,20
Each
Chemical Identifiers
CAS202982-63-6
IUPAC Name(4-chloro-2-fluorophenyl)methanaminium
Molecular FormulaC7H8ClFN
InChI KeyODMMHGMIZYLHNN-UHFFFAOYSA-O
SMILES[NH3+]CC1=CC=C(Cl)C=C1F
View more
Specifications Specification Sheet
Specification Sheet
Assay (Titration ex Chloride)≥96.0 to ≤104.0%
Appearance (Color)White to cream
FormPowder and/or lumps
Melting Point (clear melt)239-247?C
4-Chloro-2-fluorobenzylamine hydrochloride It is used as a primary and secondary intermediate. It is also used as a pharmaceutical intermediate.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
4-Chloro-2-fluorobenzylamine hydrochloride It is used as a primary and secondary intermediate. It is also used as a pharmaceutical intermediate.
Solubility
It is soluble in water.
Notes
Keep container tightly closed in a dry and well-ventilated place. Keep away from oxidizing agents. Stable under recommended storage conditions.
4-Chloro-2-fluorobenzylamine hydrochloride It is used as a primary and secondary intermediate. It is also used as a pharmaceutical intermediate.
Solubility
It is soluble in water.
Notes
Keep container tightly closed in a dry and well-ventilated place. Keep away from oxidizing agents. Stable under recommended storage conditions.
RUO – Research Use Only
General References:
- J Staroń.; D Warszycki.; J Kalinowska-Tłuścik.; G Satała. Rational design of 5-HT 6 R ligands using a bioisosteric strategy: synthesis, biological evaluation and molecular modelling. RSC Advances. 2015, 5,(33), 25806-25815.
- A Carrër.; JD Brion.; S Messaoudi.; M Alami. Palladium (II)-Catalyzed Oxidative Arylation of Quinoxalin-2 (1 H)-ones with Arylboronic Acids. Organic letters. 2013, 15,(21), 5606-5609.