Search
Thermo Scientific™
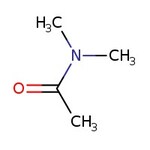
N,N-dimetilacetamida, 99 %, Thermo Scientific Chemicals
CAS: 127-19-5 | C4H9NO | 87.12 g/mol
Número de catálogo A10924.AP
Precio (EUR)
27,60
Each
Cantidad:
500 mL
Precio (EUR)
27,60
Each
Identificadores químicos
CAS127-19-5
IUPAC NameN,N-dimethylacetamide
Molecular FormulaC4H9NO
InChI KeyFXHOOIRPVKKKFG-UHFFFAOYSA-N
SMILESCN(C)C(C)=O
Ver más
Especificaciones Hoja de especificaciones
Hoja de especificaciones
CommentMaterial sourced in UK and US
Identification (FTIR)Conforms (UK-sourced material)
Refractive Index1.4375-1.4405 @ 20?C (UK-sourced material)
FormLiquid
Appearance (Color)Clear colorless
Ver más
N,N-Dimethylacetamide is used as a polar solvent in organic synthesis as well as in reactions involving strong bases such as sodium hydroxide. It is also used as a solvent for fibers like polyacrylonitrile, spandex, as an excipient in drugs viz. vumon, busulfex and in the adhesive industry. It finds application as a reaction medium in the production of pharmaceuticals, agrochemicals, dyes and plasticizers. It is widely used in polymer industry due to its high solving power for high molecular-weight polymers and synthetic resins. It plays an important role as a catalyst in various reactions viz. cyclization, halogenation, cynidation, alkylation and dehydrogenation and increases the yield of main products. It is also used as an extracting agent for oil and gases, in paint removers, in the production of photo-resist stripping compounds and as booster solvent in coatings.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Aplicaciones
La N,N-dimetilacetamida se utiliza como disolvente polar en síntesis orgánica, así como en reacciones que involucran bases fuertes como hidróxido de sodio. También se utiliza como disolvente para fibras como poliacrilonitrilo, spandex, como excipiente en drogas como vumon, busulfex y en la industria del adhesivo. Encuentra aplicación como medio de reacción en la producción de productos farmacéuticos, agroquímicos, colorantes y plastificantes. Se utiliza ampliamente en la industria de polímeros debido a su alta capacidad de resolución para polímeros de alto peso molecular y resinas sintéticas. Desempeña un papel importante como catalizador en diversas reacciones, como la ciclización, la halogenación, la cinidación, la alquilación y la deshidrogenación, y aumenta el rendimiento de los principales productos. También se utiliza como agente de extracción de aceite y gases, en limpiadores de pintura, en la producción de compuestos de desforrado fotoresistentes y como disolvente de refuerzo en recubrimientos.
Solubilidad
Miscible con agua, benceno, alcohol, acetona, éter, ésteres y cetonas.
Notas
Higroscópico. Incompatible con agentes oxidantes fuertes.
La N,N-dimetilacetamida se utiliza como disolvente polar en síntesis orgánica, así como en reacciones que involucran bases fuertes como hidróxido de sodio. También se utiliza como disolvente para fibras como poliacrilonitrilo, spandex, como excipiente en drogas como vumon, busulfex y en la industria del adhesivo. Encuentra aplicación como medio de reacción en la producción de productos farmacéuticos, agroquímicos, colorantes y plastificantes. Se utiliza ampliamente en la industria de polímeros debido a su alta capacidad de resolución para polímeros de alto peso molecular y resinas sintéticas. Desempeña un papel importante como catalizador en diversas reacciones, como la ciclización, la halogenación, la cinidación, la alquilación y la deshidrogenación, y aumenta el rendimiento de los principales productos. También se utiliza como agente de extracción de aceite y gases, en limpiadores de pintura, en la producción de compuestos de desforrado fotoresistentes y como disolvente de refuerzo en recubrimientos.
Solubilidad
Miscible con agua, benceno, alcohol, acetona, éter, ésteres y cetonas.
Notas
Higroscópico. Incompatible con agentes oxidantes fuertes.
RUO – Research Use Only
General References:
- Dipolar aprotic solvent, cf N,N-Dimethyl formamide, A13547, Dimethyl sulfoxide, A13280, 1-Methyl-2-pyrrolidinone, A12260.
- See also Sodium hydride, 13431, for warning.
- Useful solvent in halex fluorinations of chloroarenes; compare Sulfolane, A13466.
- Undergoes Vilsmeier-type reactions (compare DMF), with reactive substrates, introducing an acetyl group: Chem. Ber., 97, 616 (1964).
- Reacts with alkyl and aryl Grignard reagents to give methyl ketones in good yields: Synthesis, 228 (1984).
- In the presence of triflic anhydride and 2,4,6-collidine, forms a highly-reactive keteniminium triflate, which undergoes a [2+2] cycloaddition reaction with terminal alkenes to give, after hydrolysis a 3-alkylcyclobutanone: J. Am. Chem. Soc., 107, 2192 (1985); Org. Synth. Coll., 8, 306 (1993):
- Messaadi, A.; Salhi, H.; Das, D.; Alzamil, N. O.; Alkhaldi, M. A.; Ouerfelli, N.; Hamzaoui, A. H. A novel approach to discuss the viscosity Arrhenius behaviour and to derive the partial molar properties in binary mixtures of N, N-dimethylacetamide with 2-methoxyethanol in the temperature interval (from 298.15 to 318.15) K. Phys. Chem. Liq. 2015, 53 (4), 506-517.
- Shokouhi, M.; Farahani, H.; Jenab, M. H.; Jalili, A. H. Solubility of Hydrogen Sulfide in N-Methylacetamide and N, N-Dimethylacetamide: Experimental Measurement and Modeling. J. Chem. Eng. Data 2015, 60 (3), 499-508.