Search
Thermo Scientific Chemicals
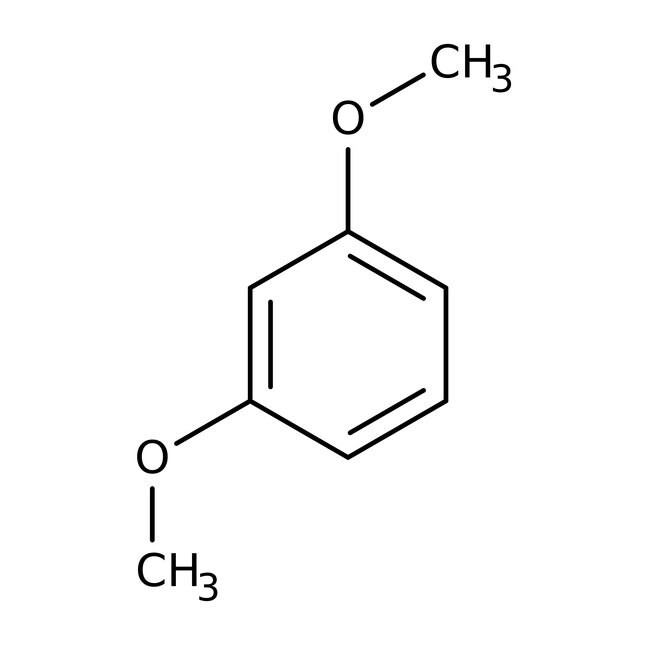
1,3-Dimetoxibenceno, 98 %, Thermo Scientific Chemicals
CAS: 151-10-0 | C8H10O2 | 138.17 g/mol
Número de catálogo A13380.36
también denominado A13380-36
Precio (EUR)
92,30
Each
Cantidad:
500 g
Precio (EUR)
92,30
Each
Identificadores químicos
CAS151-10-0
IUPAC Name1,3-dimethoxybenzene
Molecular FormulaC8H10O2
InChI KeyDPZNOMCNRMUKPS-UHFFFAOYSA-N
SMILESCOC1=CC(OC)=CC=C1
Ver más
Especificaciones Hoja de especificaciones
Hoja de especificaciones
Appearance (Color)Clear colorless to pale yellow
Assay from Suppliers CofA≥97.5%
FormLiquid
1,3-Dimethoxybenzene is used for the preparation of oxathiane spiroketal donors. It is also used for the formation of pi- and O-ylidic complexes with dichlorocarbene. Further, it acts as a flavoring agent.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Aplicaciones
El 1,3-dimetoxibenceno se utiliza en la preparación de los donantes espiroacetales de oxatiano. También se utiliza para la formación de complejos pi- y O-ilídicos con diclorocarbeno. Además, actúa como un agente aromatizante.
Solubilidad
Miscible con tolueno.
Notas
Incompatible con agentes oxidantes fuertes.
El 1,3-dimetoxibenceno se utiliza en la preparación de los donantes espiroacetales de oxatiano. También se utiliza para la formación de complejos pi- y O-ilídicos con diclorocarbeno. Además, actúa como un agente aromatizante.
Solubilidad
Miscible con tolueno.
Notas
Incompatible con agentes oxidantes fuertes.
RUO – Research Use Only
General References:
- Is readily metallated at the 2-position by n-BuLi, giving a lithio-derivative which is doubly-stabilized by oxygen coordination. For reviews, see: Org. React., 8, 258 (1954); 26, 1 (1979). The 2-lithio-derivative undergoes silylation, and the resulting silyl derivative reacts with electrophiles to give various 2-substituted products. For examples, see: Tetrahedron, 49, 10843 (1993):
- Although direct dilithiation with n-BuLi/TMEDA was unsuccessful, a sequence involving monolithiation, silylation and further lithiation at the 4-position gave the 2,4-disilyl derivative in 47% yield: J. Org. Chem., 49, 4657 (1984). For a review of the use of organosilicon intermediates in unusual electrophilic substitutions, see: Synlett, 171 (1993).
- Wang, Q.; Qi, Z.; Xie, F.; Li, X. Lewis Acid-Catalyzed Electrophilic Trifluoromethylthiolation of (Hetero)Arenes. Adv. Synth. Catal. 2015, 357 (2-3), 355-360.
- Tran, P. H.; Hansen, P. E.; Nguyen, H. T.; Le, T. N. Erbium trifluoromethanesulfonate catalyzed Friedel-Crafts acylation using aromatic carboxylic acids as acylating agents under monomode-microwave irradiation. Tetrahedron Lett. 2015, 56 (4), 612-618.