Search
Thermo Scientific Chemicals
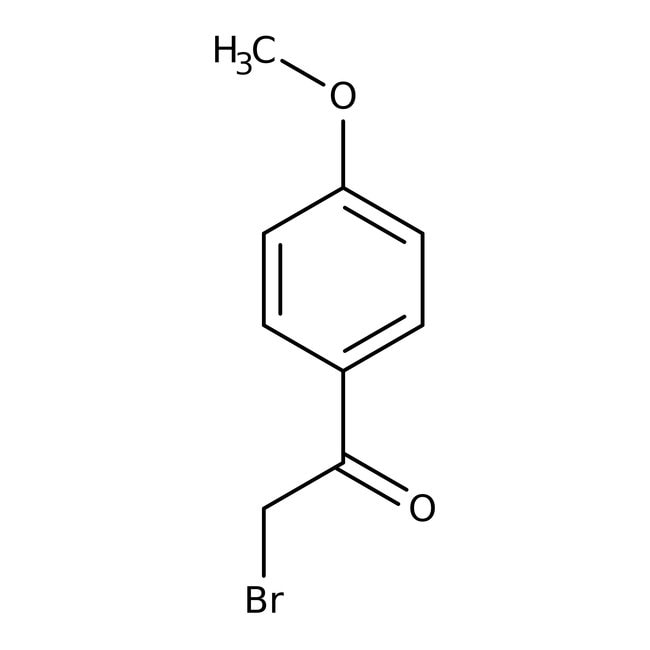
2-Bromo-4'-metoxiacetofenona, 98 %, Thermo Scientific Chemicals
CAS: 2632-13-5 | C9H9BrO2 | 229.07 g/mol
Número de catálogo A13415.22
también denominado A13415-22
Precio (EUR)
342,00
Each
Cantidad:
100 g
Precio (EUR)
342,00
Each
Identificadores químicos
CAS2632-13-5
IUPAC Name2-bromo-1-(4-methoxyphenyl)ethan-1-one
Molecular FormulaC9H9BrO2
InChI KeyXQJAHBHCLXUGEP-UHFFFAOYSA-N
SMILESCOC1=CC=C(C=C1)C(=O)CBr
Ver más
Especificaciones Hoja de especificaciones
Hoja de especificaciones
FormCrystals or powder or crystalline powder
Identification (FTIR)Conforms
Assay (GC)≥97.5%
Appearance (Color)White to cream to pale brown
Melting Point (clear melt)67-73?C
2-Bromo-4'-methoxyacetophenone, is used as a cell-permeable, covalent and potent protein tyrosine phosphatase inhibitor.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Aplicaciones
El 2-bromo-4′-metoxiacetofenona se utiliza como un inhibidor de la tirosina fosfatasa permeable a las células, covalente y potente.
Solubilidad
Soluble en DMSO, agua (parcialmente miscible), la mayoría de los disolventes orgánicos y metanol.
Notas
Almacenar en un lugar fresco. Mantenga el recipiente bien cerrado en un lugar seco y bien ventilado. Temperatura de almacenamiento recomendada: de 2 a 8 °C
El 2-bromo-4′-metoxiacetofenona se utiliza como un inhibidor de la tirosina fosfatasa permeable a las células, covalente y potente.
Solubilidad
Soluble en DMSO, agua (parcialmente miscible), la mayoría de los disolventes orgánicos y metanol.
Notas
Almacenar en un lugar fresco. Mantenga el recipiente bien cerrado en un lugar seco y bien ventilado. Temperatura de almacenamiento recomendada: de 2 a 8 °C
RUO – Research Use Only
General References:
- W Zhang.; DP Curran.; CHT Chen. Use of fluorous silica gel to separate fluorous thiol quenching derivatives in solution-phase parallel synthesis. Tetrahedron letters. 2002, 58,(20), 3871-3875.
- Y Nishiyama.; A Kobayashi. Synthesis of 1, 4-diketones: reaction of α-bromo ketones with tetrakis (dimethylamino) ethylene (TDAE). Tetrahedron letters. 2006, 47,(31), 5565-5567.
- Reagent for protection of carboxylic acids as 4-methoxyphenacyl esters, which may be prepared in the presence of KF as base: Synthesis, 897 (1981) and can be cleaved photolytically: J. Org. Chem., 38, 3771 (1973). Peptide bonds are generally unaffected by irradiation but some cleavage of trityl protecting groups may occur. See also Appendix 6. For further discussion of this and other photo-removable protecting groups, see: Synthesis, 1 (1980); Org. Photochem., 9, 225 (1987).