Search
Thermo Scientific Chemicals
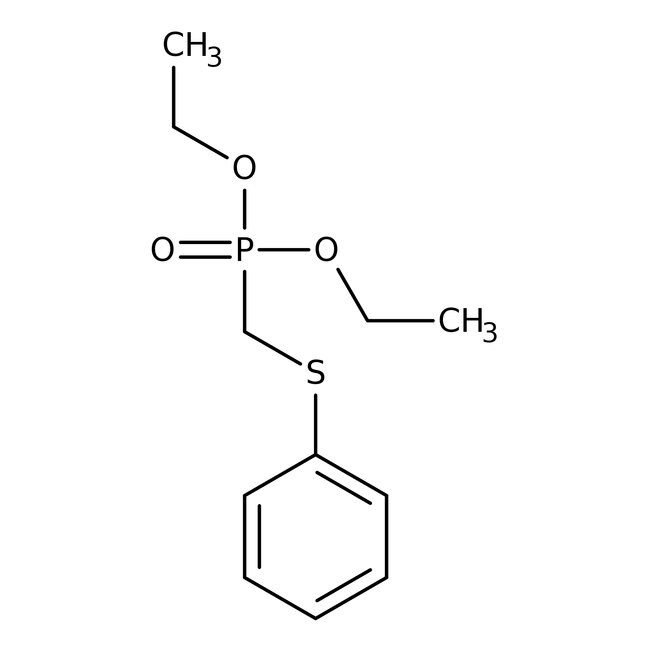
Feniltiometilfosfonato de dietilo, 96 %, Thermo Scientific Chemicals
CAS: 38066-16-9 | C11H17O3PS | 260.288 g/mol
Número de catálogo A13660.18
también denominado A13660-18
Precio (EUR)
338,00
Each
Cantidad:
50 g
Precio (EUR)
338,00
Each
Identificadores químicos
CAS38066-16-9
IUPAC Namediethyl [(phenylsulfanyl)methyl]phosphonate
Molecular FormulaC11H17O3PS
InChI KeyFBUXEPJJFVDUFE-UHFFFAOYSA-N
SMILESCCOP(=O)(CSC1=CC=CC=C1)OCC
Ver más
Especificaciones Hoja de especificaciones
Hoja de especificaciones
Appearance (Color)Clear colorless to pale yellow
FormLiquid
Refractive Index1.5310-1.5370 @ 20?C
Assay (GC)≥95.0%
Diethyl phenylthiomethylphosphonate is used to produce Diethylphosphorylmethylphenylsulfoxid, Chemical reagents, pharmaceutical research, pharmaceutical intermediates.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Aplicaciones
El feniltiometilfosfonato de dietilo se utiliza para producir dietilfosforilmetilfenilsulfóxido, reactivos químicos, intermediarios farmacéuticos y en la investigación farmacéutica.
Solubilidad
Difícil de mezclar.
Notas
Almacenar en condiciones frescas y secas en recipientes bien sellados. Mantener el recipiente herméticamente cerrado.
El feniltiometilfosfonato de dietilo se utiliza para producir dietilfosforilmetilfenilsulfóxido, reactivos químicos, intermediarios farmacéuticos y en la investigación farmacéutica.
Solubilidad
Difícil de mezclar.
Notas
Almacenar en condiciones frescas y secas en recipientes bien sellados. Mantener el recipiente herméticamente cerrado.
RUO – Research Use Only
General References:
- Horner-Wadsworth-Emmons olefination (see Appendix 1) with carbonyl compounds gives enol thioethers, convertible to aldehydes: J. Chem. Soc., 1324 (1963). Compare Diethyl methyl thiomethyl phosphonate, L00579.
- For use in a synthesis of vinylphosphonates, in which the double bond is introduced by sulfoxide elimination, see: Synthesis, 917 (1982):