Search
Thermo Scientific Chemicals
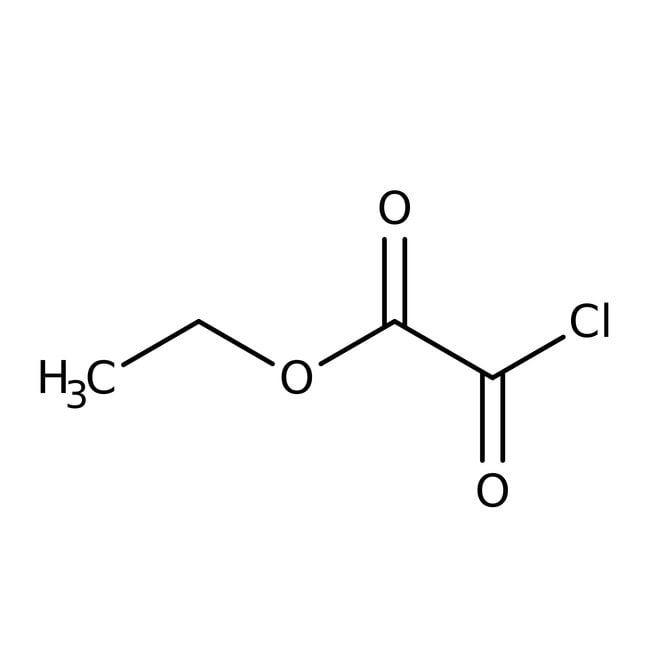
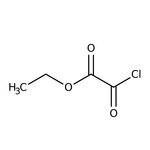
Cloruro de oxalilo etílico, 98 %, Thermo Scientific Chemicals
CAS: 4755-77-5 | C4H5ClO3 | 136.531 g/mol
Precio (EUR)
345,00
Each
Identificadores químicos
CAS4755-77-5
IUPAC Nameethyl oxalochloridate
Molecular FormulaC4H5ClO3
InChI KeyOWZFULPEVHKEKS-UHFFFAOYSA-N
SMILESCCOC(=O)C(Cl)=O
Ver más
Especificaciones Hoja de especificaciones
Hoja de especificaciones
Refractive Index1.4150-1.4190 @ 20°C
FormLiquid
Assay (GC)≥97.5%
Appearance (Color)Clear colorless to pale yellow
Identification (FTIR)Conforms
Ethyl oxalyl chloride is used in the synthesis of 2-oxo-3-alkenoic esters and alpha-keto ester in good yield. It acts as a reactant in the preparation of oxyoxalamide derivatives as an epoxide hydrolase inhibitor.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Aplicaciones
El cloruro de oxalilo etílico se utiliza en la síntesis de ésteres 2-oxo-3-alquenoicos y alfa-cetoésteres en buena producción. Actúa como reactante en la preparación de derivados de oxioxalamida como inhibidor de la epóxido hidrolasa.
Solubilidad
Ligeramente miscible con agua.
Notas
Sensible a la humedad. Incompatible con bases, agentes oxidantes y alcoholes.
El cloruro de oxalilo etílico se utiliza en la síntesis de ésteres 2-oxo-3-alquenoicos y alfa-cetoésteres en buena producción. Actúa como reactante en la preparación de derivados de oxioxalamida como inhibidor de la epóxido hidrolasa.
Solubilidad
Ligeramente miscible con agua.
Notas
Sensible a la humedad. Incompatible con bases, agentes oxidantes y alcoholes.
RUO – Research Use Only
General References:
- Reaction with imidazole, followed by a Grignard reagent, provides a route to ɑ-keto esters, generally in good yield: J. Org. Chem., 46, 211 (1981).
- In the presence of pyridine, electron-rich alkenes give 2-oxo-3-alkenoic esters: Synthesis, 137 (1986):
- Zang, T. N.; Zhao, R. R.; Yang, X. Z.; Gao, Y.; Wang, G. K.; Zhou, Y.; Zhang, J. F. A near-infrared chemodosimeter with Pi-selective colorimetric and fluorescent sensing and its application in vivo imaging. RSC Adv. 2015, 5 (88), 71756-71759.
- Monascal, Y.; Maldonado, A.; Mora, J. R.; Córdova, T.; & Chuchani, G. Homogeneous and unimolecular gas-phase thermal decomposition kinetics of methyl benzoylformate: experimental and theoretical study. J. Phys. Org. Chem. 2015, 28 (1), 40-46.